Professor — Inorganic, Organometallic, and Catalysis
Research and/or Creative Works
Research in the Waterman group applies the synthesis of novel inorganic and organometallic systems to define new reactivity and catalysis. Students will have the opportunity to prepare and fully characterize new complexes through a variety of spectroscopic techniques (e.g., NMR, IR, UV-vis, EPR), X-ray crystallography, and analytical methods. These complexes are frequently air-sensitive, and students learn to manipulate the complexes using high-vacuum and Schlenk techniques or in a glovebox. These complexes will target the catalysis of chemically important processes such as bond-forming catalysis and selective oxidation reactions. A few examples of our work follow.
A key reaction in element-element bond formation is dehydrocoupling, where two molecules with element-hydrogen bonds formally exchange E–H bonds to form E–E and H–H bonds. Dehydrocoupling is a very clean and efficient reaction taking often commercially available starting materials and providing element-element bonds with hydrogen (H2) as the only byproduct. However, for many elements this reaction is not facile, and a catalyst is needed. We have considered dehydrocoupling as a route to new molecules and materials.
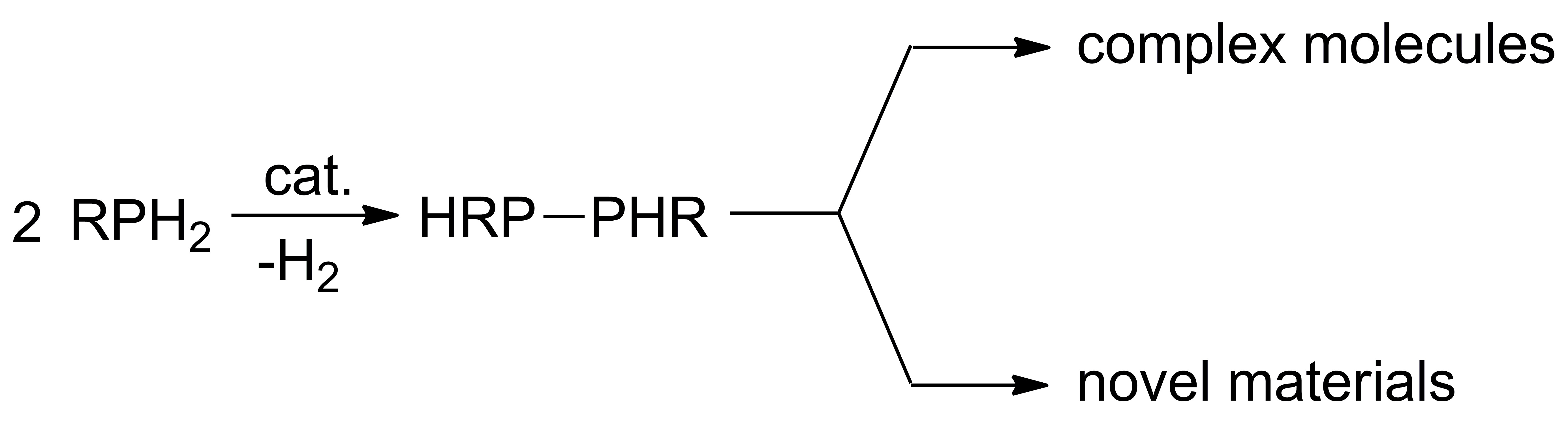
We have focuses on catalysts featuring earth-abundant metals such as cobalt, iron, tin, and zirconium among others, and their reactivity in dehydrocoupling has led to other bond-forming reactions such as hydrophosphination, or the addition of a P–H bond across an unsaturated substrate. Hydrophosphination has some particularly challenge, but we have and success in addressing some of those challenges. For example, we have identified hydrophosphination catalysts that add two substrates to alkynes and directly afford value-added diphosphinoethane products.
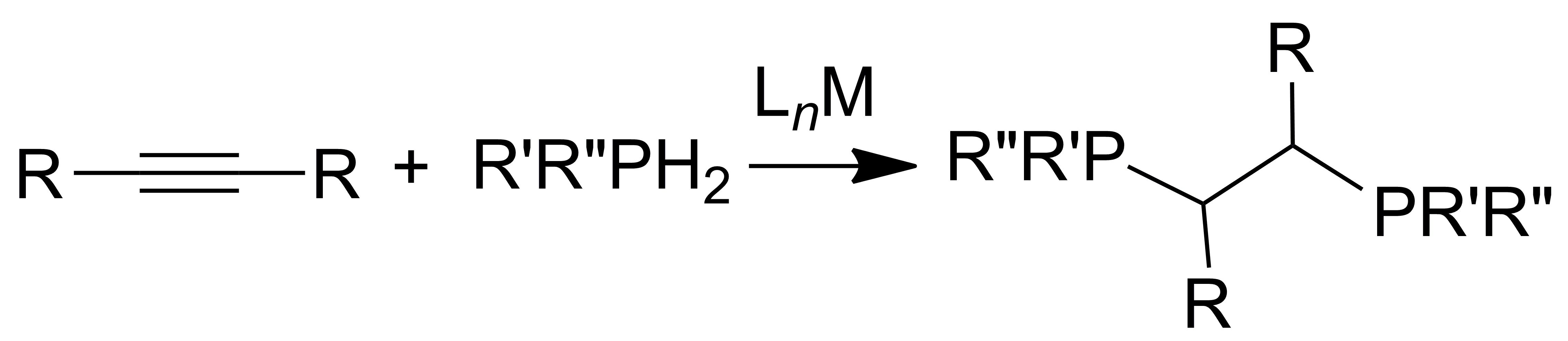
Using this knowledge we are applying this system to catalyze other element-element bond formation reactions. The predictive power of prior mechanistic study allowed us to demonstrate that other bond formation reactions are possible. For example, and in a simple periodic analogy, we have shown, for the first time, that catalytic dehydrocoupling of arsines is possible. Our continued efforts in this area are directed at addressing questions in molecular synthesis, materials science, and energy.
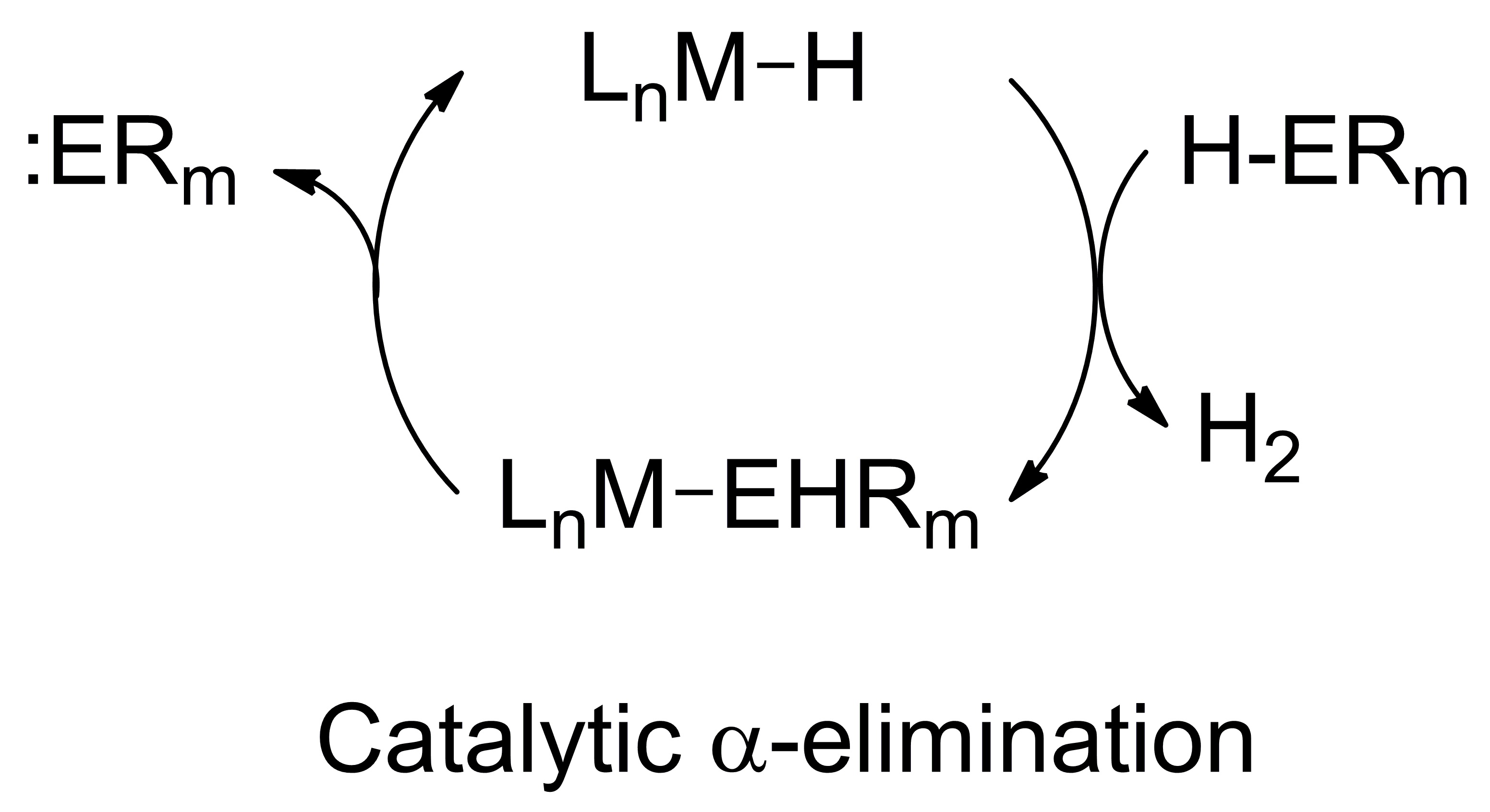
We are also interested in developing alternative routes to accessing low-valent main-group fragments such as our preparation of a phosphaalkene by insertion of an isocyanide into a zirconium-phosphorus bond. In this reaction we take a commercial available phosphorus source, a primary phosphine, and access a phosphinidene (":PR") fragment with perfect atom economy. More recently, we have identified examples of a-elimination, where a low-valent main group fragment (:ERm) is extruded from an transition-metal (LnM) catalyst, as depicted in the proposed catalytic cycle (below), which work for phosphorus and silicon. This reaction has seen limited attention in the literature, and we seek to exploit it for bond-forming catalysis.
Publications
- Seth, Jr., D. M.; Waterman, R. Photo-Initiated Radical Hydrophosphination at Titanium Compounds Capable of Ti–P Insertion Organometallics 2023, ASAP.
- Reuter, M. B; Seth, J., D. M.; Javier-Jiménez, D. R.; Finfer, E.; Beretta, E. A.; Waterman, R. Recent Advances in Catalytic Pnictogen Bond Forming Reactions via Dehydrocoupling and Hydrofunctionalization Chem. Commun. 2023, 59, 1258–1273. A 2023 Chem Comm HOT Article
- Dannenberg, S. D.; Seth, Jr. S. M.; Finfer, E. J.; Waterman, R. Divergent Mechanistic Pathways for Copper(I) Hydrophosphination Catalysis; Understanding that Allows for Diastereoselective Hydrophosphination of a Tri-substituted Styrene ACS Catalysis 2023, 13, 550–562.
- Novas, B. T.; Waterman, R. Metal-Catalyzed Hydrophosphination ChemCatChem 2022, 14, e202200988.
- Novas, B. T.; Morris, J. A.; Liptak, M. D.; Waterman, R. Effect of Photolysis on Zirconium Amino Phenoxides for the Hydrophosphination of Alkenes: Improving Catalysis Photochem 2022, 2(1), 77-87.
- Reuter, M. B.; Hageman, K.; Waterman, R. Silicon–Nitrogen Bond Formation via Heterodehydrocoupling and Catalytic N–Silylation Chem. Eur. J. 2021, 27, 3251–3261.
- Dannenberg, S. G.; Waterman, R. A Bench-Stable Catalyst for the Rapid Hydrophosphination of Activated and Unactivated Alkenes Chem. Commun. 2020, 56, 14219–14222.
- Pagano, J. K.; Xie, J.; Erickson, K. A.; Cope, S. K.; Scott, B. L.; Wu, R.; Waterman, R.; Morris, D. E.; Yang, P.; Gagliardi, L.; Kiplinger, J. L. Actinide 2-metallabiphenylenes that satisfy Hückel’s rule Nature 2020, 578, 563–567.
- Seth, Jr., D. M.; Waterman, R. Photo-Initiated Radical Hydrophosphination at Titanium Compounds Capable of Ti–P Insertion Organometallics 2023, ASAP.
- Reuter, M. B; Seth, J., D. M.; Javier-Jiménez, D. R.; Finfer, E.; Beretta, E. A.; Waterman, R. Recent Advances in Catalytic Pnictogen Bond Forming Reactions via Dehydrocoupling and Hydrofunctionalization Chem. Commun. 2023, 59, 1258–1273. A 2023 Chem Comm HOT Article
- Dannenberg, S. D.; Seth, Jr. S. M.; Finfer, E. J.; Waterman, R. Divergent Mechanistic Pathways for Copper(I) Hydrophosphination Catalysis; Understanding that Allows for Diastereoselective Hydrophosphination of a Tri-substituted Styrene ACS Catalysis 2023, 13, 550–562.
- Novas, B. T.; Waterman, R. Metal-Catalyzed Hydrophosphination ChemCatChem 2022, 14, e202200988.
- Novas, B. T.; Morris, J. A.; Liptak, M. D.; Waterman, R. Effect of Photolysis on Zirconium Amino Phenoxides for the Hydrophosphination of Alkenes: Improving Catalysis Photochem 2022, 2(1), 77-87.
- Reuter, M. B.; Hageman, K.; Waterman, R. Silicon–Nitrogen Bond Formation via Heterodehydrocoupling and Catalytic N–Silylation Chem. Eur. J. 2021, 27, 3251–3261.
- Dannenberg, S. G.; Waterman, R. A Bench-Stable Catalyst for the Rapid Hydrophosphination of Activated and Unactivated Alkenes Chem. Commun. 2020, 56, 14219–14222.
- Pagano, J. K.; Xie, J.; Erickson, K. A.; Cope, S. K.; Scott, B. L.; Wu, R.; Waterman, R.; Morris, D. E.; Yang, P.; Gagliardi, L.; Kiplinger, J. L. Actinide 2-metallabiphenylenes that satisfy Hückel’s rule Nature 2020, 578, 563–567.
Click here for more publications (link to the Waterman Group webpage)
Awards and Recognition
Japan Society for the Promotion of Science Research Fellowship, 2022
Fellow, American Association for the Advancement of Science, 2020
Elected member, Vermont Academy of Science and Engineering, 2019
Fellow, American Chemical Society, 2019
Leadership Enrichment And Development (LEAD) Award, 2017
Fellow, American Institute of Chemists, 2016
Fellow, Royal Society of Chemistry, 2015
Alexander von Humboldt Research Fellowship for Experienced Researchers, 2013
UVM Faculty Sustainability Fellow, 2010
Research Corporation Cottrell Scholar Award, 2009
Alfred P. Sloan Foundation Research Fellowship, 2009
Nominee, UVM Kroepsch-Maurice Award, 2009 & 2014
U.S. National Science Foundation CAREER Award, 2008

Areas of Expertise and/or Research
Synthetic and mechanistic inorganic/organometallic chemistry, catalysis, energy, professional development
Education
- B.S., University of Rochester, 1999
- Ph.D., University of Chicago, 2004
- Postdoctoral fellowship, University of California, Berkeley, CA (Miller Research Fellow), 2004–2006
Contact
- (802) 656-0278
Innovation Hall E334
Courses Taught
CHEM 31 — General Chemistry I
CHEM 40 — Introduction to Research
CHEM 75 — Global Energy Prospective
CHEM 131 — Inorganic Chemistry
CHEM 181 — 2nd-Year Seminar: Writing
CHEM 231 — Advanced Inorganic Chemistry
CHEM 234 — Organometallic Chemistry
CHEM 295 — Inorganic Synthesis Laboratory
