Professor and Department Chair — Organic Chemistry, Synthesis, Medicinal Chemistry
Research and/or Creative Works
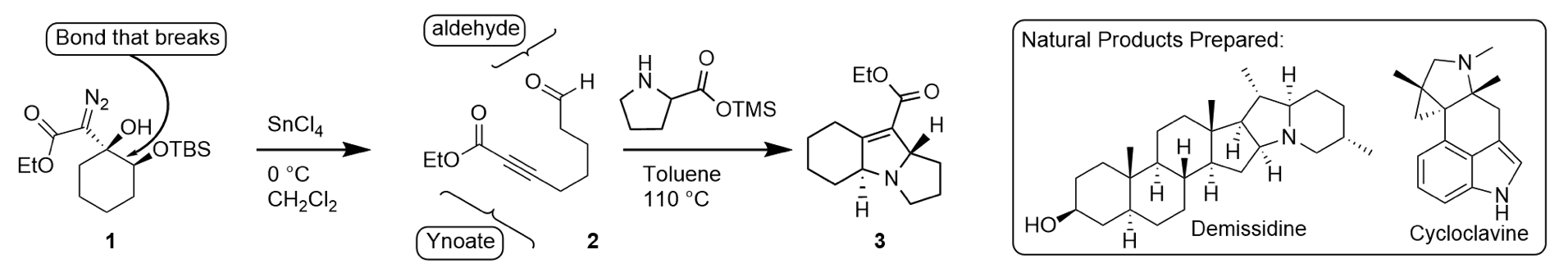
Research in our group focuses on developing new organic reactions and using these reactions to synthesize structurally complex organic molecules including natural products and non-natural medicinal agents. To achieve this, we have studied the reactivity of several highly unstable intermediates including vinyl cations and heteroallene salts. In both cases, these highly reactive intermediates have allowed us to develop surprising and useful reactions. For example, our work with vinyl cations led us to discover a convenient ring fragmentation reaction that provides two synthetically useful functional groups (a carbon-carbon triple bond in conjugation with a carbonyl (i.e. an ynoate) and an aldehyde; 1 to 2, Scheme 1). These products are useful because they can be taken advantage of in further synthetic transformations including intramolecular 1,3-dipolar cycloadditions to give structurally-complex nitrogen-containing heterocyclic compounds (e.g. 2 to 3, Scheme 1). Nitrogen-containing heterocycles are ubiquitous in natural products and medicinal agents and we have used this methodology in the total synthesis of two unrelated alkaloids (demissidine and cycloclavine).
Scheme 1
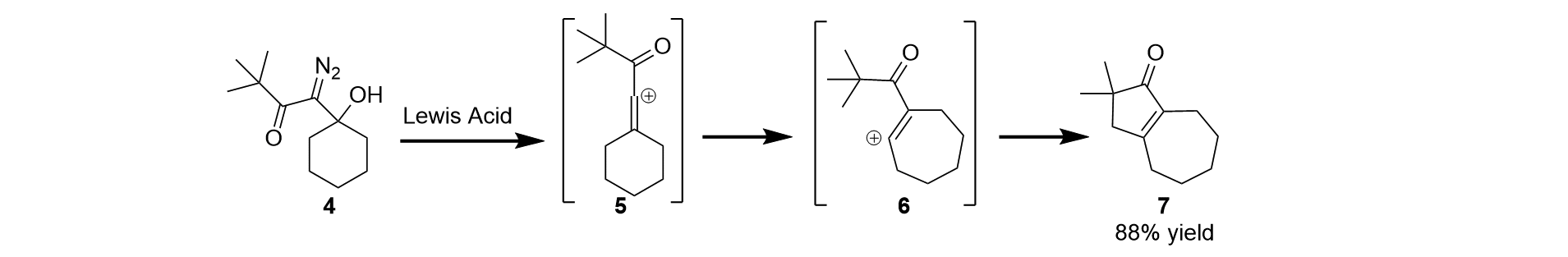
We have also studied the ability of vinyl cations to react in intramolecular C-H insertion reactions. Insertion reactions are powerful because they can convert a typically inert C-H bond into a new C-C bond, which allows for new synthetic approaches that are greener and more efficient. In this vein, we have discovered that β-hydroxy-α-diazo ketones (e.g. 4, Scheme 2) react with Lewis acids to give vinyl cation intermediates that rearrange (e.g. 5 to 6) and then undergo C-H insertion to give cyclopentenone products (e.g. 7) in good yield.
Scheme 2
My research group has also been studying intramolecular reactions of 1-aza-2-azoniaallene salts (Figure 1). These studies have led us to discover that these high-energy heteroallene intermediates can react by multiple mechanistically-distinct pathways including (3 + 2) cycloadditions, (4 + 2) cycloadditions, (2 + 1) cycloadditions, and C-H insertions to afford structurally diverse heterocyclic scaffolds (Figure 1). This is important from a fundamental reactivity standpoint (we have shown that these intermediates can react in unprecedented ways), and also from a practical application standpoint (the products formed are structurally unique and could be useful either for biological studies or as synthetic intermediates).
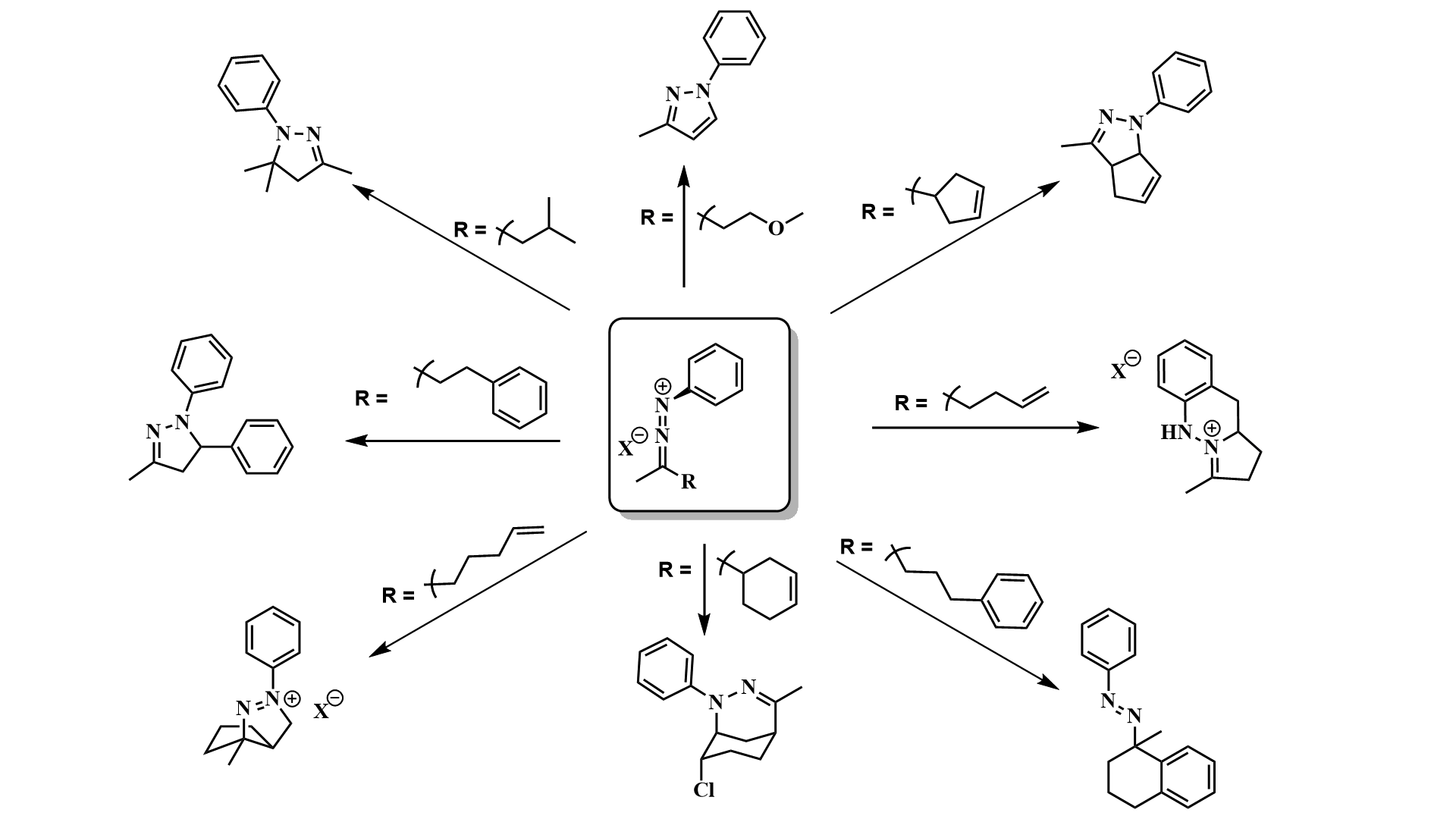
Figure 1. 1-Aza-2-azoniaallene salts can undergo a variety of mechanistically-distinct intramolecular reactions.
Finally, my research group is part of a cross-college collaboration with Prof. Victor May (Dept. of Neurological Sciences) and Prof. Jianing Li (Department of Chemistry) that aims to discover small molecule antagonists of the PAC1 receptor as therapeutics for anxiety related disorders including PTSD. Animal studies have shown that pituitary adenylate cyclase activating polypeptide (PACAP) and its receptor (PAC1) are involved in stress and anxiety related disorders, and PACAP blood levels are elevated in female patients who suffer from PTSD. Importantly, a truncated form of PACAP (PACAP6-38), a known antagonist of PAC1, blunts stress- and pain-induced anxiety in animal models. Unfortunately, PACAP6-38 is not a viable drug candidate.
Prof. May is an expert in the field of cellular and molecular functions of PACAP signaling and the behavioral roles of PACAP in stress-mediated anxiety-related disorders, and Prof. Li is an expert in computational protein modeling with experience in modeling protein binding sites. Prof. Li has recently identified two potential binding sites within the PAC1 receptor. With this knowledge in hand, my group has begun synthesizing a series of compounds that will be tested by Prof. Victor May for their ability to act as PAC1 receptor antagonists. Through iterative cycles of design, synthesis and testing we hope to ultimately develop therapeutic strategies for stress and anxiety management.
Publications
Click here for more publications (link to the Brewer Group webpage)
S. E. Cleary, X. Li, L.-C. Yang, K. N. Houk, X. Hong, M. Brewer, "Reactivity Profiles of Diazo Amides, Esters and Ketones in Transition Metal Free C-H Insertion Reactions" J. Am. Chem. Soc. 2019, 141(8), pp 3558–3565.
J. Fang, M. Brewer, "Intramolecular Vinylation of Aryl Rings by Vinyl Cations" Org. Lett. 2018, 20(23), 7384-7387.
R. Dhakal, M. Ivancic, M. Brewer, “Two-Step Sequence of Cycloadditions Gives Structurally Complex Tetracyclic 1,2,3,4-Tetrahydrocinnoline” J. Org. Chem. 2018, 83(11), 6202-6209.
S. Cleary, M. Hensinger and M. Brewer, “Remote C-H Insertion of Vinyl Cations Leading to Cyclopentenones” Chem. Sci. 2017, 8, 6810–6814.
C. Liao, X. Zhao, M. Brewer, V. May and J. Li, “Conformational Transitions of the Pituitary Adenylate Cyclase-Activating Polypeptide Receptor, a Human Class B GPCR” Sci. Rep. 2017, 7, 5427–5434.
N. Al-Bataineh, K. N. Houk, M. Brewer, X. Hong, “(2+1)-Cycloaddition Reactions Give Further Evidence of the Nitrenium-like Character of 1-Aza-2-azoniaallene Salts” J. Org. Chem. 2017, 82, 4001–4005.
G. M. Giampa, J. Fang, M. Brewer, “A Route to the C, D, E Ring System of the Aspidosperma Alkaloids” Org. Lett. 2016, 18, 3952–3955.
R. C. Dhakal, M. Brewer, “Intramolecular (4+2) Cycloaddition of Aryl-1-aza-2-azoniaallene Salts: a Practical Approach to Highly Sterically-congested Polycyclic Protonated Azomethine Imines” Tetrahedron 2016, 72, 3718–3728.
X. Hong, D. Bercovici, Z. Yang, N. Al-Bataineh, R. Srinivasan, R. Dhakal, K. N. Houk, M. Brewer, "Mechanism and Dynamics of Intramolecular C-H Insertion Reactions of 1-Aza-2-azoniaallene Salts" J. Am. Chem. Soc. 2015, 137, 9100–9107.
X. Hong, Y. Liang, M. Brewer, K. N. Houk, “How Tethers Control the Chemo- and Regio-Selectivities of Intramolecular Cycloadditions between Aryl-1-Aza-2-Azoniaallenes and Alkenes” Org. Lett. 2014, 16, 4260–4263.
A. Bayir, M. Brewer, “Fragmentation of Bicyclic γ-Silyloxy-β-hydroxy-α-diazolactones as an Approach to Ynolides” J. Org. Chem. 2014, 79, 6037–6046.
D. A. Bercovici, J. M. Ogilvie, N. Tsvetkov, M. Brewer, “Intramolecular Polar [4 + 2]-Cycloadditions of Aryl-1-aza-2-azoniaallene Salts: Unprecedented Reactivity Leading to Polycyclic Protonated Azomethine Imines” Angew. Chem. Int. Ed. 2013, 52, 13338–13341.
D. A. Bercovici, M. Brewer, “Stereospecific Intramolecular C–H Amination of 1-Aza-2-azoniaallene Salts”, J. Am. Chem. Soc. 2012, 134, 9890–9893.
Awards and Recognition
Thieme Chemistry Journals Award, 2009
NSF CAREER Award, 2008
Amgen New Faculty Award, 2006
Amgen New Faculty Award, 2005
NIH National Research Service Award Postdoctoral Research Fellowship, 2002
APS Bruce W. Erickson Young Investigator Award (2nd Place), 2001

Areas of Expertise and/or Research
Organic chemistry, synthetic methods, natural products synthesis, synthesis of small molecule medicinal agents
Education
- Ph.D., University of Wisconsin, Madison, WI, 2002
- Postdoctoral Fellowship, University of California, Irvine, CA, 2002–2005
Contact
- (802) 656-1042
Innovation Hall E343 and E222
