Professor – Inorganic Chemistry
Research and/or Creative Works
Current thinking is that over a quarter of all proteins require a metal. These metalloproteins have diverse biological functions, including: oxygen transport, electron transfer, and enzyme catalysis. The Liptak group is particularly interested in elucidating the mechanisms of metalloenzymes that rely upon transition metals such as iron, cobalt, and nickel. Currently, the major thrust of the research group is unravelling the mechanisms of heme iron acquisition by pathogenic bacteria to lay the foundation for future antibiotic development. A second research focus of the group is revealing the mechanisms of metal tetrapyrrole biosynthesis with the long-term goal of designing synthetic enzymes for production of catalysts with alternative energy applications.
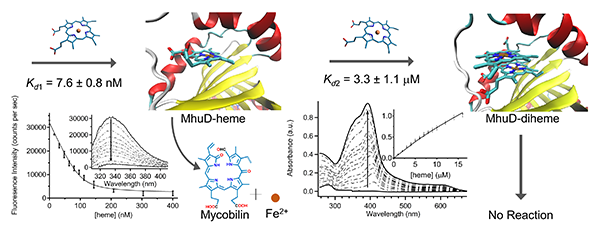
Mycobacterium tuberculosis MhuD can bind one or two heme molecules
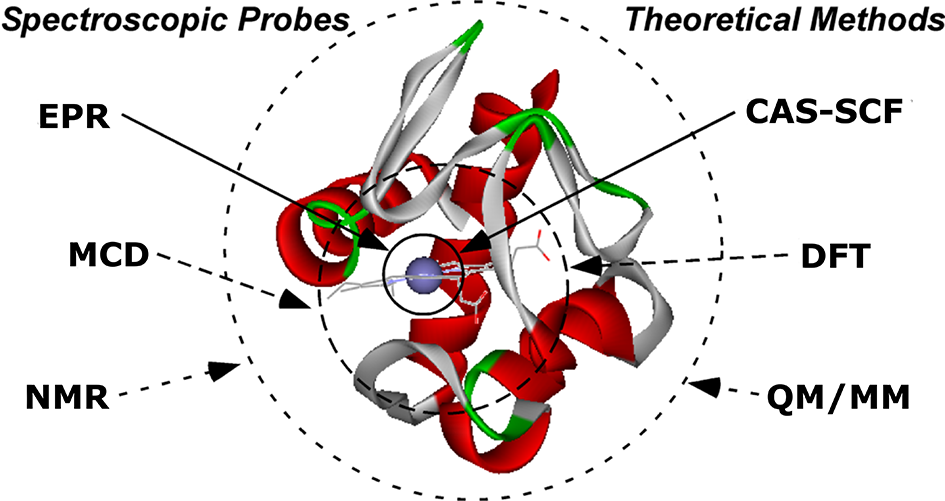
One of the major challenges for understanding metalloenzyme function is the role of electronic structure. Transition metals have the unique ability to adopt numerous oxidation, spin, and configurational states under ambient conditions and each of these electronic states has unique reactivity. In other words, simply knowing which metal is bound to an enzyme is not enough to predict its chemical reactivity! The Liptak group has developed diverse biochemical (recombinant protein expression, purification, and air-sensitive sample handling), spectroscopic (fluorescence -> electron paramagnetic resonance; time-dependent density functional theory -> hybrid quantum mechanics / molecular mechanics.), as well as computational (density functional theory, time-dependent density functional theory, and multi-reference perturbation theory) expertise to address these complex problems.
Researchers in the Liptak group learn a variety of biochemical, spectroscopic, and computational skills in addition to management of a research project from the first experiments to journal publication. As a result, graduate student alumni have secured jobs in both academia and industry. The majority of undergraduate alumni have moved on to graduate programs in chemistry.
Publications
Click here for more publications (link to the Liptak Group webpage)
- Johnson, K.L.; Graves, A.G.; Eckhert, K.; Liptak, M.D. "Second-sphere tuning of analogues for the ferric-hydroperoxoheme form of Mycobacterium tuberculosis MhuD" J. Inorg. Biochem. 2023, 246, 112300. DOI:10.1016/j.jinorgbio.2023.112300
- Petrucci, A.N.; Cousins, M.E.; Liptak, M.D. "Beyond 'Mega': Origin of the 'Giga" Stokes shift for triazolopyridiniums" J. Phys. Chem. B 2022, 126, 6997-7005. DOI:10.1021/acs.jpcb.2c04397
- Morris, J.A.; Lickey, B.S.; Liptak, M.D. “Insertion of cobalt into tetrapyrroles" Vitam. Horm. 2022, 119, 1-22. DOI:10.1016/bs.vh.2022.01.013
- Schuelke-Sanchez, A.E.; Cornetta, A.R.; Kocian, T.A.J.; Conger, M.A.; Liptak, M.D. “Ruffling is essential for Staphylococcus aureus IsdG-catalyzed degradation of heme to staphylobilin" J. Inorg. Biochem. 2022, 230, 111775. DOI:10.1016/j.jinorgbio.2022.111775
Thakuri, B., O'Rourke, B.D., Graves, A.B., Liptak, M.D. "A dynamic substrate is required for MhuD-catalyzed degradation of heme to mycoblilin" Biochemistry. 2021, 60, 918-928.
A. E. Schuelke-Sanchez, A. A. Stone, M.D. Liptak. "CfbA promotes insertion of cobalt and nickel into ruffled tetrapyrrole in vitro" Dalton Trans. 2020, 49, 1065-1076.
M. A. Conger, A. R. Cornetta, M.D. Liptak. "Spectroscopic evidence for electronic control of heme hydroxylation by IsdG" Inorg. Chem. 2019, 58, 15455-15465.
B. Shao, N. Stankewitz, J. A. Morris, M. D. Liptak, I. Aprahamian. "White-light emission from a structurally simple hydrazone" Chem. Comm. 2019, 55, 9551-9554.
B. Thakuri, A. B. Graves, A. Chao, S. L. Johansen, C. W. Goulding, M. D. Liptak. "The affinity of MhuD for heme is consistent with a heme degrading function in vivo" Metallomics 2018, 10, 1560-1563.
M. A. Conger, D. Pokhrel, M. D. Liptak. “Tight Binding of Heme to Staphylococcus aureus IsdG and IsdI Precludes Design of a Competitive Inhibitor” Metallomics 2017, 9, 556–563.
H. Qian, M. E. Cousins, E. H. Horak, A. Wakefield, M. D. Liptak, I. Aprahamian. “Suppression of Kasha’s Rule (SOKR): A Novel Mechanism to Explain Aggregation Induced Emission” Nature Chem. 2017, 9, 83–87.
A. B. Graves, E. H. Horak, M. D. Liptak. “Dynamic Ruffling Distortion of the Heme Substrate in Non-Canonical Heme Oxygenase Enzymes” Dalton Trans. 2016, 45, 10058–10067.
A. B. Graves, M. T. Graves, M. D. Liptak. “Measurement of Heme Ruffling Changes in MhuD Using UV/Vis Spectroscopy” J. Phys. Chem. B 2016, 120, 3844–3853.
C. L. Lockhart, M. A. Conger, D. S. Pittman, M. D. Liptak. “Hydrogen Bond Donation to the Heme Distal Ligand of Staphylococcus aureus IsdG Tunes the Electronic Structure” J. Biol. Inorg. Chem. 2015, 20, 757–770.
A. B. Graves, R. P. Morse, A. Chao, A. Iniguez, C. W. Goulding, M. D. Liptak. “Crystallographic and Spectroscopic Insights into Heme Degradation by Mycobacterium tuberculosis MhuD” Inorg. Chem. 2014, 53, 5931–5940.
Awards and Recognition
New Talent: Americas, 2016
Paul Saltman Award, 2015
Ruth Kirschstein-NRSA, 2009
Barry M. Goldwater Scholarship, 2002

Areas of Expertise and/or Research
Bioinorganic chemistry, magneto-optical spectroscopy, computational chemistry
Education
- B.A., Hamilton College, Clinton, NY, 2003
- Ph.D., University of Wisconsin, Madison, WI, 2008
- NIH NRSA Postdoctoral Research Fellow, University of Rochester, Rochester, NY, 2008–2011
Contact
- (802) 656-0161
Innovation E336
Courses Taught
Chem 031 — General Chemistry 1 (Fall 2015, Spring 2017)
Chem 131 — Inorganic Chemistry (Spring 2018, 2020)
Chem 231 — Advanced Inorganic Chemistry (Fall 2011-2014, 2016-2019)
Chem 236 — Physical Inorganic Chemistry (Spring 2012, 2014, 2016, 2019)
