Water Quality
Long Term Acid Sensitive Lake Monitoring at the Lye Brook Wilderness Area

During the 1980s, initial chemical surveys were conducted on lakes throughout Vermont. Concern was mounting that remote, high-elevation lakes in geologically sensitive areas were acidified or risked acidification due to the long distance transport of atmospheric pollution. Initial monitoring within the southern Green Mountains indicated that this region of Vermont was especially sensitive to acidification and that a high proportion of the undeveloped lakes were notably acidic. In 1993, the VMC partnered with VT DEC to continue monitoring in the Lye Brook Wilderness on Branch and Bourn Ponds. Acid lake monitoring in Vermont is funded by the US EPA’s Long Term Monitoring Program (LTM), which supports VTDEC’s work to sample 12 acidic lakes throughout Vermont, including Bourn and Branch Ponds.
The Data
Long-term water chemistry monitoring has been carried out at two acid-sensitive lakes in the Lye Brook Wilderness area since the 1980s. The ponds, Bourn and Branch Ponds, are approximately 9.0 and 12.0 meters deep respectively, dark in color, with low pH and thermally stratify in the summer. Samples are collected three times a year in the spring, summer and fall using a Kemmerer water sampler. The samples are collected at 1 m below the surface and 1-2 m from the bottom in the summer when the lake is stratified. Measurements include field pH, lab pH, secchi transparency, temperature, true color, apparent color, gran alkalinity, specific conductance, as well as concentrations of calcium, magnesium, sodium, potassium, aluminum (both speciated monomeric, and total dissolved), nitrate, sulfate, chloride and dissolved organic carbon (DOC). Branch is more acidic than Bourn and is located just south of the Lye Brook Wilderness boundary. The methods of collection, processing and analysis have remained consistent for nearly 30 years. Last year’s analysis focused on measurements from the epilimnion, the uppermost layer in a thermally stratified lake. This year, the focus will be on the deeper area of the lake, the hypolimnion, typically between 1-2 meters off the bottom at the deep hole.
2015 in Summary
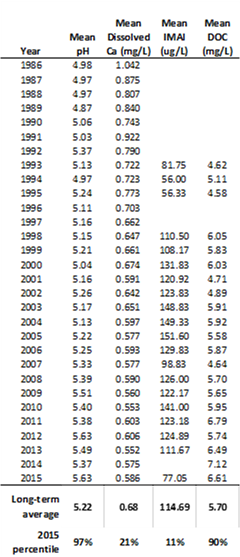
In 2015, water quality trends in the epilimnion (top layer) of Bourn and Branch Ponds followed the same patterns observed in 2014, and over the long-term record. This includes low concentrations of both base cation (Ca, Mg, Na, K) and acid anions (SO4, NO3 and Cl). The decline in acid anion concentrations can be correlated to federally mandated reductions in national pollution emissions and deposition. Mean pH, alkalinity and dissolved organic carbon values continue to be some of the highest on record (Figure 33, Table 9). This is consistent with acid lake measurements made across the Northeast, showing reduced acidity (higher alkalinity), lower toxic inorganic monomeric aluminum (IMAL) and increased DOC.
Long-Term Trends
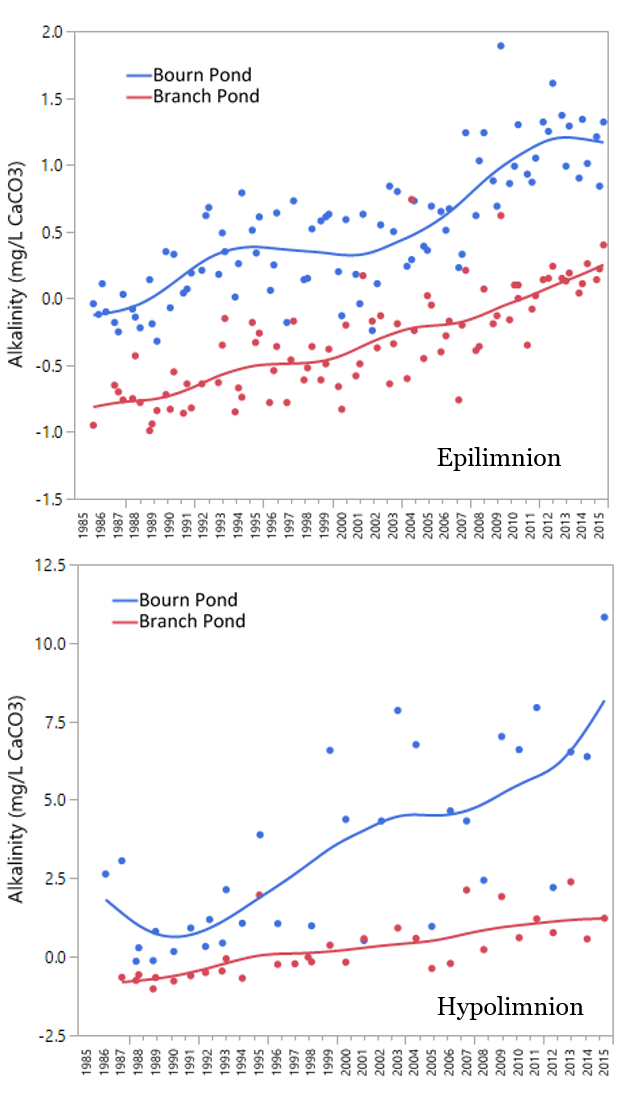
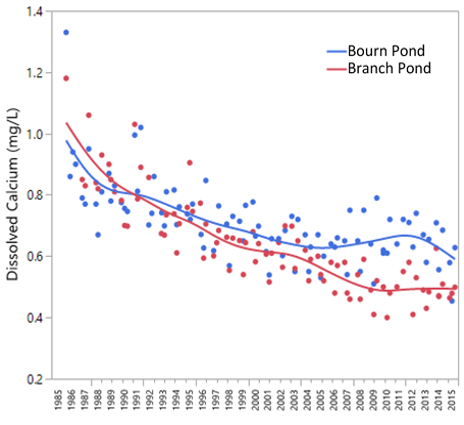
There are several notable trends in Vermont’s acid lakes. First we’ll discuss the top layer of the pond, the epilimnion. Alkalinity, a measure of a waterbodies’ acid buffering capacity is now positive on both Bourn and Branch Ponds. This is most striking on the more acidic Branch Pond which has yielded positive alkalinities since 2011 (Figure 33, top). Calcium has declined by half since the 1980s but appears to be levelling off since the mid-2000s (Figure 34). The toxic form of aluminum, Inorganic Monomeric Aluminum (IMAL) is declining on both ponds. This is a beneficial trend for fish reproduction and survival. The phenomena of increased DOC is called lake browning and is occurring across the northeastern US, eastern Canada and northern Europe (Lawrence 2013). It has many possible ecological consequences including greater oxygen depletion and changes to the food web structure (Williamson et al, 2015). Some possible causes for brownification include increased organic matter solubility in soils (Monteith et al. 2007, SanClements et al. 2012), increased CO2 in the atmosphere, increased precipitation and reduced atmospheric inputs. No conclusive cause has been identified thus far.
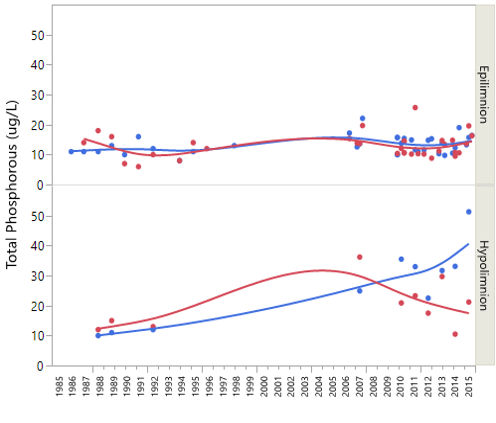
In the summer, samples collected from Bourn Pond’s hypolimnion during stratification smell like “rotten eggs” due to the lack of oxygen (anoxia) and buildup of hydrogen sulfide (H2S) that’s released from lake sediments. The anoxic conditions in the deepest area of the lake allow for oxidation or chemical reducing conditions to occur. This causes SO4 to reduce to sulfide (as in hydrogen sulfide - H2S) near the lake sediments. When the hypolimnion sample is decanted at the surface the smell is strong. With thermal stratification, water chemistry in the hypolimnion can be dramatically different from spring to summer to fall. During the summer, anoxic conditions lead to the release of phosphorus, and temporary increases pH and alkalinity. This combined with the long-term reduction of sulfur and nitrogen oxides has resulted in a long-term improvement in the hypolimnion alkalinity (Figure 33 top). Phosphorus concentrations in the hypolimnion are nearly twice that of the epilimnion. This may be due to internal loading from sediments. Under certain conditions, phosphorus in sediments may be released due to warmer water, low dissolved oxygen, and changes in pH (Figure 35).
Biological improvements are the bottom line for determining recovery in acid-impaired lakes. Some changes on Bourn and Branch Ponds are beneficial while others will hamper recovery. Recovery of biota will be limited by low concentrations of base cations, specifically calcium, a nutrient essential to the development and reproduction of fish and macroinvertebrates. Inorganic monomeric aluminum is declining, reducing the toxicity and harm to biota. Paired with improvements in pH and alkalinity, the waters are becoming less acidic and less toxic. Ironically, the increases in pH and microbial activity may increase the phosphorus loading to these otherwise low nutrient lakes. Dissolved organic carbon levels have been increasing at both Bourn and Branch Ponds (Figure 36), which could aid in biological recovery through increased microbial food availability and reducing bioavailability of certain metals. Specifically, DOC mediates the impacts of the toxic inorganic monomeric aluminum to biota, and can speed conversion of toxic aluminum to organic forms beyond what is expected solely from decreases in acid deposition (Lawrence et al. 2013). Ironically, the increases in pH and microbial activity may increase the phosphorus loading to these otherwise low nutrient lakes.
Implications
Federal mandates under the Clean Air Act require Class I Wilderness Areas (like Lye Brook) to protect air-quality and related ecosystem values. The data collected at these waterbodies provides one line of evidence with which to monitor and document the ecological impacts of both atmospheric pollution, and the policies meant to limit such impacts.

Acid-sensitive lakes have improved significantly since the 1990 Clean Air Act Amendments. But, because of the loss of calcium and magnesium throughout the watershed and in the lakes, sensitive fish, snails, and insect species will not recover until further reductions in sulfur and nitrogen occur. Increases in dissolved organic carbon will aid in biological recovery as this weak organic acid binds with the toxic form of aluminum. Bedrock and soils need time to resupply calcium and magnesium to the lakes through the weathering process.
Analysis of air and water quality by means of chemical and biological inventories on these sensitive lakes will be maintained. Monitoring the lakes in Lye Brook Wilderness can be used to document trends resulting from emission reductions required by the Clean Air Act, but also to identify any potential new sources of stress or pollution, such as climate change. Such indicator trends are being used to inform management targeted towards the recovery of these water bodies.

While improvements in the acidity of high elevation surface waters are promising, a legacy of decreased base cations and increased toxic aluminum levels indicate that biological recovery remains uncertain.
References
- SanClements, M.D., G.P. Oelsner, D.M. McKnight, J.L. Stoddard and S.J. Nelson. 2012. New Insights into the Source of Decadal Increases of Dissolved Organic Matter in Acid Sensitive Lakes of the Northeastern United States. Environmental Science & Technology 46(6):3212-3219.
- Williamson, C.E., Overholt, E.P., Pilla, R.M., Leach, T.H., Brentrup, J.A., Knoll, L.B, Mette, E.M and Moeller, R.E. 2015. Ecological consequences of longterm browning in lakes. Scientific Reports 5. 186666.
- Monteith, D.T., J.L. Stoddard, C.D. Evans, H.A. de Wit, M. Forsius, T. Hogasen, A. Wilander, B. Skjelkvale, D.S. Jeffries, J. Vuorenmaa, B. Keller, J. Kopacek, and J. Vesely. 2007. Dissolved organic carbon trends resulting from changes in atmospheric deposition chemistry. Nature 450(7169): 537-540.
- Lawrence, G.B, J.E. Dukett, N. Houck, P. Snyder, and S. Capone. 2013. Environmental Science and Technolology. 47 (13): 7095–7100
Additional Resources
- Vermont Monitoring Programs for Acid Rain: http://www.watershedmanagement.vt.gov/bass/htm/bs_acidrain-mon.htm
- US Environmental Protection Agency Long Term Monitoring Program: http://www2.epa.gov/airmarkets/monitoring-surface-water-chemistry
VMC Project Database Link
- Biological And Chemical Survey Of Selected Surface Waters In Lye Brook Wilderness Area: Water Chemistry Of Water Bodies: https://www.uvm.edu/femc/project/long-term-monitoring-acid-sensitive-lakes