1.
COMMITTEE’S
MISSION
The
University of Vermont is committed to the humane care and use of
animals in
activities related to research, testing and teaching. The University
has
adopted the animal care principles in accordance with the Guide
for the Care and Use of Laboratory
Animals
(“the
Guide”),
and in compliance with applicable federal, state, and local laws and
regulations, such as the federal Animal Welfare Act, and Public Health
Service
Policy on Humane Care and Use of Laboratory Animals (PHS).
The
University of Vermont Institutional Animal Care and Use Committee
(IACUC) has
an Assurance on file with the Office of Laboratory Animal Welfare in
accordance
with the PHS Policy. The Committee is regulated by the USDA under the
Animal
Welfare Act as documented in the Code of Federal Regulation Title 9,
Subchapter
A Parts 1, 2 and 3. The
University’s
Animal Care and Use Program is fully accredited by AAALAC International.
Assuring
laboratory animal welfare necessitates a partnership among the
Institutional
Official (IO), the IACUC, the University Veterinarian and the
investigators. Ultimately,
accountability for assuring humane care and use of the animals resides
with the
institution, but this may only be achieved when all the constituents
contribute
to this shared goal. The
following
organizational chart clearly outlines the direct lines of
responsibility and
corresponding authority.
|
|
|
|
|
University
of Vermont and State Agricultural College Board of Trustees |
|
|
|
|
|
|||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||||
|
|
|
|
|
PRESIDENT (CEO) |
|
|
|
|
|
|||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||||
|
|
VICE
PRESIDENT FOR RESEARCH Authorized Institutional Official |
|
||||||||||||||||||||||||
|
|
|
|
|
|||||||||||||||||||||||
|
|
|
|
||||||||||||||||||||||||
|
VETERINARIAN Office
of Animal Care
Management |
|
Priority
One Services, Inc. (Contract
Management) |
|
Research
Protections Office |
|
Staff Support |
|
INSTITUTIONAL
ANIMAL
CARE & USE COMMITTEE (IACUC) |
|
|||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||
|
College
of Arts
& Sciences |
|
College
of Medicine |
|
College
of Engineering &
Mathematical Sciences |
|
College
of Agriculture
& Life
Sciences |
|
College
of Nursing & Health Sciences |
|
Rubenstein
School of Environment & Natural Resources |
|
|
||||||||||||||
|
|
||||||||||||||||||||||||||
In
order to accomplish the objectives inherent in these regulations and
principles, there are primarily two organizational components
designated to
ensure their implementation in the overall animal care and use program.
1.
The Institutional Animal Care and Use Committee (IACUC) is the
University’s
central review body for matters relating to the care, use and treatment
of
animals in these areas. The IACUC office is located within the Research
Protections Office.
2.
The Office of Animal Care Management is responsible for oversight of
all animal
care and use and for ensuring compliance with federal, state and local
regulations. The University Veterinarian is the Director of this office.
3. Priority One Services,
Inc. is the contract
manager for the Animal Resources Center.
2. COMMITTEE’S RESPONSIBILITIES/AUTHORITY/COMPOSITION
The
IACUC was established in accordance with the Animal Welfare Act and the
Health
Research Extension Act under the authority of the Vice President for
Research
and Dean of the Graduate College to ensure the humane care and use of
animals
for research and education at the University under optimum conditions,
which,
at a minimum, comply with all pertinent laws.
The
Office of Animal Care Management (OACM)/Animal Resources Center (ARC)
is
administered by the University Veterinarian/Priority One Services, Inc.
and is
charged with the veterinary care and husbandry of the animals, the
occupational
health and safety of personnel, and ensuring the appropriate training
of
personnel working with animals in accordance with all relevant
regulations and
guidelines governing the humane care and use of animals. The University
Veterinarian and the OACM are under the authority of the Vice President
for Research
and Dean of the Graduate College.
2.A. Responsibilities and Authority
1.
Review and report at least
once every six
months, on the evaluation of the research facility’s program for the
humane
care and use of animals.
2.
Inspect,
evaluate and report to the Institutional
Official, at least once every six months, inspection findings of the
institution’s
animal facilities, including satellite facilities and animal study
areas.
3.
Review and investigate
legitimate concerns
involving the care and use of animals at the institution.
4.
Make
written recommendations to the Institutional Official regarding any
aspect of
the research, animal program, facilities or personnel training.
5.
Review
and have authority to approve, require modifications in (to secure
approval),
withhold approval of those sections of applications or proposals
related to the
care and use of animals.
6.
Review
and have authority to approve, require modifications in (to secure
approval) or
withhold approval of proposed significant changes regarding the use of
animals
in ongoing activities.
7.
Notify
investigators and the institution in writing of its decision to approve
or
withhold approval of those activities related to the care and use of
animals,
or of modifications required to secure IACUC approval as set forth in
the PHS
Policy at IV.C.4.
8.
Conduct
continuing review of each previously approved, ongoing activity covered
by PHS
Policy at appropriate intervals as determined by the IACUC, including a
complete
review in accordance with the PHS Policy at IV.C. 1-4. at least once
every
three years.
9.
Be
authorized to suspend an activity involving animals as set forth in the
PHS
Policy at IV.C.6.
2.B. Composition
Committee
membership is comprised of members with varying professional and
personal
backgrounds and who have demonstrated a genuine interest in and
commitment to
the purpose of the Committee. Membership includes one Doctor of
Veterinary
Medicine, one practicing scientist, one member whose primary concerns
are in a
nonscientific area, one individual who is not affiliated with the
institution
in any way other than as a member of the IACUC.
3. CONTACTS
The administrative
office of the IACUC is
located in 213
Waterman Bldg, 85
South Prospect St, Burlington, VT 05405, (802) 656-5040. The RPO staff as well as a
list of the
current Committee Chairs and the Veterinarian is located under contacts
on our website.
4. OFFICE OF ANIMAL CARE MANAGEMENT (OACM)
As
stated above, the University Veterinarian administers the Office of
Animal Care
Management (OACM), which is charged with the veterinary care and
husbandry of
the animals, the occupational health and safety of personnel, and
ensuring the
appropriate training of personnel working with animals in accordance
with all
relevant regulations and guidelines governing the humane care and use
of
animals.
Further
information is available at web site http://www.uvm.edu/~oacm/
The
main OACM office is located in 116 Hills Building. Administrative
personnel are
as follows:
Dr.
Ruth Blauwiekel, University Veterinarian 656-0459 drruth@uvm.edu
John
Lovelette, Business Manager 656-2206
Project
Manager, Priority One Services, Inc., 656-1006 facmgr@med.uvm.edu
5. TRAINING REQUIREMENTS AND OCCUPATIONAL HEALTH
A
web-based training program, Collaborative
Institutional Training Initiative (CITI),
is used to provide training and
incorporates local content. All Principle Investigators and key
personnel must
take the “General Lab Animal Training – Basic Course” through CITI in
addition
to the species specific course for each species
that is listed on the
protocol before they will be added to the roster. Training is required
to be
completed every three years.
·
General
lab animal training:
IACUC procedures, the
principles of the Three R’s, methods for minimizing animal pain and
distress,
facility access and logistics, use of PPE, basic animal observation and
restraint, reporting animal welfare concerns, surgery/anesthesia,
euthanasia,
blood collection, social housing etc.
·
Species
specific courses: Species specific course(s)
must be taken for
each species that is listed on the protocol on which you are key
personnel. Species
courses include; mouse, rat,
amphibians, guinea pigs, rabbits, cattle, horses, and swine.
IACUC
staff members
have created a training
resource and
FAQ page
which includes a link to the training completion
dropdown
to confirm that personnel
have completed the relevant modules prior to addition to the protocol
roster.
PIs and research staff can also access this dropdown to confirm the
completion
of courses.
Hands-on
biomethods
training is offered by the OACM Veterinary Technicians and is a
requirement
before facility access will be granted. When the University
Veterinarian
participates in training (such as for surgical procedures or confirming
competence in conditionally-acceptable methods of euthanasia), this
training is
documented and the training records for hands-on biomethods training is
maintained by the Veterinary Technicians.
Training
of Animal Care
staff includes review of relevant SOP's, one-on-one training by the
Facility Manager,
and "shadowing" of more experienced personnel. In addition, Animal
Care staff all review the IACUC and the Risk Management &
Safety on-line
training and complete the learning-assessment tools. Other
opportunities for
training include staff and animal user meetings and seminars, a
quarterly Animal Care
newsletter, and postings to
the Animal Care list-serve.
5.A.
Specific Training
5.B.1.a. Working With Hazardous
Agents in Animals
Those
involved with the use of hazardous agents/organisms are
primarily trained for working with those agents/organisms in their
research
laboratories by the PI. The
Environmental Safety Office
(EHS) monitors the use of chemical and biohazardous agents and
the Radiation
Safety Office
(RSO)
monitors radiation
exposure.
NOTE: All projects
proposing to utilize recombinant DNA or infectious agents require
review and
approval from The Institutional Biosafety Committee (IBC) prior to
IACUC
submission. The IBC
is charged with
reviewing all research projects and activities involving recombinant
DNA (as
outlined in the “Guidelines for Research Involving Recombinant DNA
Molecules”)
to assure that specific practices for constructing and handling (i)
recombinant
DNA molecules and (ii) organisms and viruses containing recombinant DNA
molecules are followed. The IBC is also charged with responsibility for
reviewing
the use of infectious agents in research at UVM. A representative of
the Office
of Environmental Safety serves on the IBC, as does the Director of the
Department of Risk Management, the University Veterinarian, the
Radiation
Safety Director, and faculty of the College of Medicine with particular
expertise in infectious agents. For
more information go to
https://www.uvm.edu/rpo/biosafety-oversight
and http://www.uvm.edu/~radsafe/.
The
EHS offers Chemical Safety Training which covers general
laboratory safety and an overview of regulations, OSHA laboratory
standards and
the UVM Chemical Hygiene plan. Training may also be provided by the
person’s
laboratory supervisor, provided that training is adequately documented.
Chemical labeling and storage requirements and a copy of the UVM
Laboratory
Safety Audit are available on the EHS website http://www.uvm.edu/safety/.
The
RSO requires that a person complete Radiation Safety Training
prior to using radioisotopes or radiography in the laboratory. A
certification
exam is required at the completion of the course. The Radiation Safety
Handbook
is available online.
All
animal caretakers are instructed and trained by the Animal
Care Facility Manager in the husbandry, sanitation procedures and
precautions
to be followed when working in any biohazard area. Each protocol
utilizing
hazardous agents requires an instruction sheet which is reviewed by the
Facility Manager with the caretakers and is posted on the animal room
door. The
instructions on the sheet include animal and personnel safety.
Refer
also to the
“Biohazardous
Materials”
policy.
5.B. Continuing Training
Ongoing
“hands-on” training for present
and incoming research personnel is provided by the veterinary staff.
Regularly
scheduled small group sessions cover restraint, injection and sampling
techniques, recognition of common health problems, aseptic technique,
anesthesia
and euthanasia. This training is continually provided to individuals on
an
as-needed basis following consultation with the University Veterinarian.
CITI
training courses are required to
be completed every three years as of November 1, 2017. Reminders
letters will
be sent to personnel as their training expiration date nears.
5.C. Other Resources
Workshops,
guest speakers and/or
consultation on various relevant topics such as research models and
mouse
colony management are provided to the research community.
Library
support for searches for
alternatives to animal use and/or procedures which cause more than
momentary or
slight pain and distress to animals is available through the University
Library
Services. The
Office of Animal Care
Management and the Research Protections Office have numerous reference
materials available.
Information
on the following topics is
also available: Levels
of
discomfort/distress in animal experimentation; anesthetic and analgesic
drug
formulary for different species; AVMA Guidelines for Euthanasia.
Self-study
materials on handling and
basic manipulative procedures for commonly used laboratory animals are
available.
Video
tapes are available on ethics, animal
uses, handling techniques, and surgery both in the library and the OACM
office.
The
Northern Mountain Branch of AALAS is a
membership association dedicated to advancing and disseminating
knowledge about
the responsible care and use of laboratory animals for the benefit of
human and
animal health. Animal Care staff are members and attend meetings, which
happen
three times per year and are open to all involved in the use of animals.
5.D. Occupational Health and Safety Program
The University's Department of Risk Management & Safety
(RM&S) has contracted with an occupational health provider, Champlain
Medical, to provide occupational
health monitoring for University personnel who work with animals. A written
risk assessment survey must be completed by each person listed on an active
IACUC protocol. This survey is routed through the Research Protections Office
(RPO) and logged before being forwarded to Champlain Medical. Access to the
animal facilities is not granted to personnel until they have completed and
returned the survey. For Animal Care workers, the medical evaluation is sent to
the Priority One occupational health provider, Concentra, for review. The
Animal Care medical evaluation may include a physical exam, pulmonary function
test and review of immunization status. Animal Care employees are
required to have a current tetanus immunization; tetanus vaccination is offered
and encouraged for other personnel. For University employees and
students, the risk assessment is used to determine who should receive
additional follow-up and remediation (which may include pulmonary function
test, use of an N95 mask, appropriate immunizations, or other precautionary
measures). An annual reassessment is required for all personnel and students
who continue to work with animals.
Zoonotic agents, animal allergies and other hazards are described
in the required web-based IACUC training for personnel and assessment of these
risks is addressed in the risk assessment form that is submitted to Champlain
Medical. The annual re-assessment form addresses changes in health status such
as pregnancy or immunocompromise. Animal Care staff members must
read and sign off on a written SOP before performing any activity that may be
hazardous and in addition are personally trained by the Facility Manager. All
research personnel who work with infectious agents sign an informed consent
document acknowledging that they are aware of the hazards and have read the relevant
SOPs. In the case of infectious agents, personnel who are
immunocompromised or women who are pregnant are informed of the risk and
advised to notify their supervisors. Minor injuries (such as animal bites,
sprains, etc.) are treated at Champlain Medical or at the University of Vermont
Medical Center. University of Vermont Medical Center emergency
services are readily accessible for more serious injuries should they occur.
Risk Management & Safety (RM&S) manages laboratory safety
and hazardous chemical waste and the use of radioisotopes on campus. RM&S
oversees the use of chemical and biological agents to ensure that they are used
safely and in accordance with all applicable government regulations and
University policies and procedures. RM&S also conducts training in the
handling of hazardous chemicals, biohazardous agents and blood-borne pathogens.
RM&S staff members conduct regular audits of each laboratory as described
in UVM's Environmental Management Plan (http://esf.uvm.edu/uvmemp/).
The University of Vermont has an Institutional Biosafety Committee
(IBC) in accordance with the NIH "Guidelines for Research Involving
Recombinant DNA Molecules" and other relevant federal
regulation. Investigators utilizing recombinant DNA and/or
infectious agents must submit a protocol to the IBC for review and approval.
The University Veterinarian may determine during pre-submission of an IACUC
protocol that a concurrent IBC application should be prepared and submitted to
the IBC. In addition, the Biosafety Officer or his designate reviews the
Hazardous Materials section of any IACUC protocol utilizing hazardous
chemicals, ionizing radiation, infectious agents or recombinant DNA, and
conducts laboratory risk assessments as appropriate. This shared membership and
communication between the IACUC and the IBC helps to ensure that risks are
recognized and managed effectively.
Animals exposed to or treated with infectious agents are
maintained in rooms with double barriers in filter-topped caging or in
semi-rigid isolators. When appropriate, a Class II Biosafety cabinet (BSCll) is
used for husbandry and other procedures. Laboratories using infectious agents
in animals must submit information about the agent and an SOP and/or BARD
(Biohazardous Agent Reference Document) for working with the animals to the
Animal Facility Manager. Containment practices and other information relative
to the specific biological agent housed in a room then are posted on the animal
room door. For chemical agents, animals' cages are marked with special cage cards and the bedding and caging are handled as appropriate
to the agent. For biological agents, bedding and other animal wastes are either
double-bagged in a BSCll and sealed/boxed for incineration or autoclaved prior
to disposal in the regular trash. All BSCll are certified annually.
Personal Protective Equipment (PPE) appropriate to the room is
indicated by signage on the door and discarded in the anteroom prior to exiting
the area. PPE is provided by the animal facility for use by research personnel
as well as animal care staff. This PPE may include isolation gowns and masks,
gloves, shoe covers, bouffant head covers, N95 respirators, face shields and
Tyvek coveralls, depending on the agent and procedures.
All research personnel who work with infectious agents must report
any accident involving exposure to the agent to their supervisor immediately
and visit the emergency room or consult with the Infectious Disease Physician
on call. A plan of action is filed with the IBC and prophylactic
antibiotics/vaccines may be inventoried in either the laboratory or the
University of Vermont Medical Center pharmacy, depending on the risk assessment
and plan for that specific agent. Serology may be done to detect changes in
antibody titer and further follow-up is conducted as deemed appropriate by the
Infectious Disease Physician. There are no nonhuman primate species or small
ruminants currently in use as models at UVM.
All animal care personnel are provided with work garments for
daily routine wear (usually scrubs, occasionally coveralls), which are
laundered by commercial arrangement (Unifirst). All animal care personnel are
provided with rubber steel-toed boots or shoes, plastic aprons, heavy rubber
gloves, ear protection, goggles, fit-tested respirators and other PPE when
required. Hands are washed upon leaving an animal holding room; each room has a
sink and antimicrobial soap available. When departing one building for another,
caretakers are required to change to street clothes and change to clean scrubs
when entering the next facility. No work-issued clothing is taken home or worn
outside the animal care areas.
Animals traveling to any laboratory or procedural space outside of
the animal facilities must be in filter-topped caging on a cart and covered by
draping material, a box, or some other secondary container. Signage
is posted in any elevators which are used for animal transport.
Exceptions: Undergraduate students whose exposure is limited to
one hour per week per semester, research involving aquatic animals, or
personnel who work only with animal products are not required to enroll in the
program. Undergraduate students are made aware of the program and
are provided with information about risks. Students in need of
attention should contact the Student Health Center. While IACUC
Board members are not required to take part in the program, they are strongly
encouraged to participate if they attend semiannual inspections.
Any other exceptions to required program enrollment are considered
on a case-by-case basis by the UVM veterinarian.
5.E. Training for Students on Teaching Protocols
Students
working on
protocols in which animal activities are limited to teaching are
required to
complete appropriate CITI trainings before they can start working with
live
animals. This
includes the “General Lab Animal Training – Basic Course” and the
species-specific
course for each species that is listed on the protocol. At
the discretion of the of the course instructor,
non-Animal Science students having limited involvement with animals may
take
only the species specific course.
The
course instructor is responsible for
ensuring completion of required training for students enrolled in the
class.
If
the role of the
student, however, involves responsibilities beyond that which is
typically
required of the class, such as husbandry or performing animal
procedures, the
student is required to take additional training as appropriate. The University
Veterinarian should be
contacted by the instructor for guidance.
6. FELLOWS, RESIDENTS, POST-DOCTORAL FELLOWS, ASSOCIATES, TRAINEES, AND STUDENTS CONDUCTING VERTEBRATE ANIMAL RESEARCH
Fellows,
residents, post-doctoral
fellows, post-doctoral associates, post-doctoral trainees, and students
(graduate or undergraduate) cannot conduct vertebrate animal research
without
having a faculty sponsor/instructor who is responsible for overseeing
the
research activities.
The faculty sponsor is responsible
for:
a) reviewing the materials for
submission to
the IACUC for accuracy and completeness;
b) assisting and supporting the
student in
his/her interaction with the IACUC and for overseeing the resolution of
any
issues arising during the review process; and
c) oversight of the student's
research to
ensure that the protocol is followed as approved.
Students who are principal
investigators have
responsibilities as listed in Investigator
Responsibilities.
7. COMMITTEE REVIEW AND SUBMISSION PROCESS
7.A. Types of Review
All
projects (with or without internal or external funding) which involve
the use
of vertebrate animals must undergo IACUC review and receive approval
prior to
initiation. Investigators are required
to consult with the veterinarian and obtain her signature on the final
protocol
form prior to submitting the protocol form to the Committee.
There are
three types of Committee review: full, designated or administrative.
Determination of the type of review is usually based upon the expected
level of
animal pain or discomfort and types of procedures.
For pain levels see policy “Determination
of Levels of Pain & Distress.”
NOTE: Any projects
proposing to utilize recombinant DNA or infectious agents, requires
review and
approval from The Institutional Biosafety Committee (IBC) prior to
IACUC
submission. The IBC
is charged with
reviewing all research projects and activities involving recombinant
DNA (as
outlined in the “Guidelines for Research Involving Recombinant DNA
Molecules”)
to assure that specific practices for constructing and handling (i)
recombinant
DNA molecules and (ii) organisms and viruses containing recombinant DNA
molecules are followed. The IBC is also charged with responsibility for
reviewing the use of infectious agents in research at UVM. A
representative of
the Office of Environmental Safety serves on the IBC, as does the
Director of
the Department of Risk Management, the University Veterinarian, the
Radiation
Safety Director, and faculty in the College of Medicine with particular
expertise in infectious agents. For
more information go to https://www.uvm.edu/rpo/biosafety-oversight
and
http://www.uvm.edu/~radsafe/.
7.A.1.
Full Committee Review
A
full committee review is required for all protocols with pain levels of
D or
higher. The IACUC
uses a
primary/secondary reviewer method for full Committee reviews. The
primary reviewer is always a scientific representative of the Committee
and is
responsible for presenting a summary of the protocol at a fully convened meeting along with any
concerns or points requiring
clarification and/or stipulations. A secondary reviewer is usually
assigned by
the IACUC Chair. The
secondary reviewer adds
any additional concerns. The University Veterinarian then outlines any
additional issues and then the review is opened for discussion by the
Committee. All
Committee members are provided relevant materials
and have access to all other documentation related to the study.
7.A.2. Designated
Review
Protocols
that meet the criteria for designated review are posted to allow all
IACUC
members an opportunity to review.
If no member
calls for a Full Committee review, the IACUC Chair will assign one
Committee
member and the University Veterinarian to complete the review. The reviewers’ comments and
clarifications
and/or stipulations are resolved with the investigator.
The Designated IACUC reviewer or the IACUC
Chair may at any point call for a Full Committee Review.
7.A.3. Administrative Review
The
chair alone (or his/her designee from the Committee) reviews and
approves actions
in this category. This category captures review of actions that do not
require
an actual Committee review, e.g., minor amendments, some continuing
reviews,
previously approved protocols that have been resubmitted or identical
protocols
submitted to different funding agencies, protocols with no direct
animal use,
or when funds will be used for salary support only on a previously
approved
protocol.
7.A.4. Veterinarian
Verification
and Consultation
The
VVC process
can be used to make significant changes to animal activities that are
part of a
protocol that was previously reviewed and approved by Full Committee
Review
(FCR) or Designated Member Review (DMR) The IACUC has granted authority
to the
University Veterinarian to verify the following changes:
·
Procedures
outlined in Guidelines for the Performance of Repeat Procedures
(Appendix 3)
·
Changes
to anesthetics, analgesics, antibiotics, or fluid replacement based on
VVC
Formulary for Rodents at UVM (Appendix 4)
·
Changes
in housing location to another location that is IACUC approved.
·
Euthanasia
methods within the AVMA guidelines for Euthanasia of Animals.
Amendments
to existing IACUC approved protocols are submitted to the IACUC for
review and
must be accompanied by an updated Animal Use Protocol. The RPO analyst
will
evaluate the submission and determine if it meets criteria for VVC. If
it does,
the analyst will electronically assign the amendment to the University
Veterinarian. Once the veterinarian verifies that the requested change
complies
with the IACUC approved policies, she will verify the change through
InfoEd.
Following the review and sign-off, the analyst will issue an approval
letter to
the PI. Changes may not be implemented until the approval letter is
issued.
If
the University Veterinarian does not believe that the amendment falls
under
VVC, she will note that in her review and the amendment will be
assigned either
to DMR or FCR as applicable.
7.A.5. Projects
in which
Animal Use is Limited to Animal Products
Animal products is defined as material obtained from a USDA slaughterhouse, animal byproducts, or shared animal products from other investigators. These projects do not require IACUC review. For additional information see the Animal Tissue policy.
7.A.6. Projects
in which
Animal Use is Limited to Teaching Activities
Protocols
in which animal use is limited to teaching activities may be submitted
using
the Teaching Protocol form in lieu of the Animal Use Protocol form.
This form
is primarily used for Animal Science projects that
are primarily or solely used for teaching purposes when pain category
is less
than C, which do not include procedures that cause more than momentary
pain or
distress.
Researchers are still required to submit teaching protocols to the
University
Veterinarian for review prior to submitting it to the Committee.
Contact RPO
staff for advice.
7.B. Criteria for Review
All
proposed activities are reviewed to ensure that the following federal
requirements
for granting IACUC approval are met:
Activities
--
All activities
involving animals must be in accordance with USDA Regulations/PHS
Policy.
Animal
numbers and
group sizes
– The IACUC requires that the experimental design be described and
animal
numbers justified either with a power calculation or a reference to
previously-published work of comparable scientific design. A
biostatistician
serves on the Committee and has the specific charge of evaluating the
numbers
justification section. An online biostatistics tutorial providing
guidance for
the investigators was launched in the spring of 2013.
Harm/benefit
analysis –
All protocols which
entail more than momentary pain/distress (USDA category D or E) are
reviewed in
a convened meeting of the IACUC. During the discussion, the IACUC notes
the
responses of the investigator to the rationale for utilizing animals
and the
potential significance of the work. For protocols which entail more
than
momentary pain/distress without provision of analgesia, a specific
scientific
justification for withholding analgesia is required. If the Committee
has
further questions regarding the harm/benefit analysis, the investigator
may be
required to provide further clarification prior to approval of the
protocol.
Search
for alternatives –
In the case of any
protocol which entails more than momentary pain or distress to the
animals, the
investigator must perform a literature review seeking alternatives
which could
achieve the same scientific objectives with a lesser degree of
pain/distress.
The investigator must describe in a narrative what leads him/her to the
conclusion that there are no alternatives to the proposed procedures.
Rationale
and Methods
-- All proposals must
include:
•Identification
of the species and the approximate number of animals to be used;
•
A rationale for involving animals and for the appropriateness of the
species
and numbers of animals to be used;
•
A complete description of the proposed use of the animals;
•
A description of procedures designed to assure that discomfort and pain
to
animals will be limited to that which is unavoidable for the conduct of
scientifically valuable research, including provision for the use of
analgesic,
anesthetic, and tranquilizing drugs where indicated and appropriate to
minimize
discomfort and pain to animals; and
•
A description of any euthanasia method to be used.
Note: Some investigators have
requested to use
videotaping as part of their protocol.
Videotaping cannot be used as a substitute for protocol
monitoring
activities and there are strict criteria about controlling access,
especially
if it is being viewed externally.
Contact the RPO for advice.
Duplication
--
Assurance that
activities do not unnecessarily duplicate previous efforts must be
provided.
Surgery
-- Requirements for
sterile surgery and pre/postoperative care must be met. An animal may
not be used
for several major operative procedures from which it will recover,
without
meeting specified conditions.
Euthanasia--The
euthanasia method
must be consistent with the recommendations of the current AVMA Panel
on
Euthanasia (2000 edition or later).
Housing/Health
--
Animal living
conditions must be consistent with standards of housing, feeding and
care
directed by veterinarian or scientist with appropriate expertise.
Medical care
must be provided by qualified veterinarian.
Qualifications
--Personnel must be
appropriately trained and qualified. Completion of all the University
of
Vermont's Office of Animal Care Management training program is required
for all
individuals working with animals or identified on a protocol.
Deviation
from
Requirements
--Must be justified for scientific reasons, in writing.
The
Committee’s review process always includes a check for compliance with
all
applicable IACUC or institutional policies and procedures.
7.C. Initial New Protocol Review and Submission Process
7.C.1. Initial
New Protocol
Review
Any project with USDA
pain level D
or above requires a
“full committee” review. (see
7.A.1.
above).
Any project with USDA pain level C or below may qualify for a “designated” review. (see 7.A.2. above)
IACUC
review efforts focus on the appropriateness of animal numbers,
procedures and
adequacy of investigator skills. Normally scientific peer review is
left to
outside funding agencies. In lieu of outside review, IACUC requires the
department of record to certify that the research project has
scientific merit by conducting an internal review. This internal review
can be done one of two ways; 1) through an internal review committee or
2) through independent scientific review which requires the sign-off of
two knowledgeable faculty members. In both cases, the Scientific
Research Plan and References (section D.4. of the Animal Use Protocol
form) must be examined. Please note that if a Faculty Sponsor is listed
on the protocol, the Faculty Sponsor should not be the one conducting
the scientific merit review.
7.C.2. Initial
New Protocol
Submission
Meetings
are routinely held on the fourth Monday of each month. The deadlines
for
submission of materials for full committee review are: the first Monday
of the
month to the Veterinarian and the second Monday of the month to the
IACUC
office. The decision as to the type of review a proposal receives
(designated
or full committee) is based on the expected level of animal discomfort
and
types of procedures. Protocols are placed on the agenda as they come in.
Step
1: Submission of
the Protocol. After consultation with the University Veterinarian and
obtaining
the veterinarian's signature, an investigator submits a completed IACUC
protocol to the IACUC Research Review Analyst.
Step
2: Initial Review.
Following the receipt of a Protocol, the type of review a proposal will
receive
(full, designated or administrative) is determined based on the
expected level
of animal pain and distress and types of procedures proposed. This
determination is usually made by the IACUC chair in consultation with
IACUC
staff. The veterinarian also may be consulted if there is a question
about the
type of review which is appropriate. Protocols designated USDA Pain
level
"D" or "E" are given full review at a convened IACUC meeting.
Completeness of the protocol also is checked by the RPO staff at this
time.
Step
3: Review (By Type). At convened monthly meetings, the IACUC considers
new
protocols requiring full committee review and reviews the Reports of
Designated
Reviews and Reports of Administrative Reviews. Possible
outcomes of the Committee's
review include unqualified Approval, Modifications Required to Secure
Initial
Approval or Withhold Approval.
7.C.3. Review of
Grants
The
IACUC is required to ensure that all
research described in a grant application or proposal is entirely
consistent
with any corresponding protocol(s) reviewed and approved by the IACUC. Any discrepancies must be
resolved prior to
the start of the project. The
IACUC
works with Pre-Award Services to establish that an appropriate
connection is
made between the application and the protocol being reviewed.
7.C.3.i. New Competing or Competing Renewal Grant Applications
To
meet the requirement listed above, any time
you will be submitting a new competing
grant or a competing renewal
application, a new protocol must
be submitted to the IACUC for review and approval.
Pre-Award Services will not clear funds for
release until there is an approved protocol.
Continued approvals will be checked on an annual basis for
the life of
the grant.
Note: It
is very important that the proposal and protocol be matched correctly
for the
reason mentioned above.
7.C.3.ii. When the Project is a New Competing or a Competing Renewal Application and the New Protocol is Identical or Substantially Similar to an Approved Protocol
Obtaining
grant funding is extremely
competitive. The
same grant proposal may
be submitted to multiple funding agencies at once or the same agency at
different
time points. If you
obtain new funding,
it is your responsibility to submit the corresponding grant and
protocol for IACUC
review and approval.
The
IACUC treats identical protocols as new
applications, however, a new committee review may not be required if
the
project is the same or substantially similar to the previously approved
protocol.
If
this is the case, you must submit the
following:
1. One
copy of your new grant application and the corresponding protocol.
2. A
letter to the committee chair explaining that you are submitting a
similar
grant application to a different funding agency.
State that this new protocol application is
identical to the old one (provide IACUC file #) with regard to
hypotheses,
specific aims, and vertebrate animal involvement (or describe minor
differences).
If
this application is essentially the same as
the previously approved application with only minor differences clearly
described in a letter, the protocol will receive administrative review. If substantial changes are
proposed, then a
new committee review may be required.
7.C.3.iii. Competing Resubmissions or Supplements
Grant
resubmissions require an amendment to a
previously approved protocol if it is identical or substantially
similar to
that protocol and grant. The
amendment
form and a copy of the resubmitted grant application are to be
submitted for
review and approval. Administrative
and
competitive supplements also require an amendment to a previously
approved
protocol. The
amendment form and a copy
of the supplement are to be submitted for review and approval.
7.C.3.iv.
Just-in-Time Provision for IACUC Submissions
What
is
“Just-in-Time” Review
The
NIH just-in-time policy allows grant
applications to be submitted to NIH for peer review without prior IACUC
approval. This
policy has been extended
by the University to all UVM grant proposals where the granting agency
does not
require IACUC approval at the time the proposal is submitted. Researchers should check
with the Pre-Award
Services Office to determine the funding agency’s IACUC approval
requirements.
Process
for “Just-in-Time” Review
If
the sponsor accepts just-in-time vertebrate
animal review, as soon as the researcher is notified that the proposal
received
a favorable priority ranking from the granting agency, the protocol
should be
submitted to the IACUC for review.
If
the project is a new or competing renewal and is
identical or substantially similar to a previously approved
protocol, see section 7.C.3.ii., above, for further guidance. If the project is not identical or substantially similar,
researchers should check
the IACUC submission deadlines for the next available IACUC meeting as
special
requests for insertion onto an agenda after the scheduled deadline may
not be
possible. NOTE: It
is not necessary for
the researcher to submit a protocol if the priority ranking is
unfavorable.
If
the just-in-time request is for a
resubmission, see section 7.C.3.iii. for submission guidance.
The
delay in submission of a protocol for IACUC
review approval may delay an award but should not affect the receipt of
an
award.
7.C.4. Interinstitutional Assurance Agreement
UVM
researchers may arrange to carry out
portions of research that involve the use of animals via an NIH-funded
subcontract from an NIH-awardee (often a private company) that does not
itself
have an approved Animal Welfare Assurance on file with the NIH Office
of
Laboratory Animal Welfare (OLAW). In such situations, OLAW requires
that the
company apply for an "Interinstitutional Assurance," under which UVM
assures that the research will be carried out under the terms of its
own
OLAW-approved Animal Welfare Assurance.
Interinstitutional
Assurance Agreements
require signatures from the private company, UVM Associate VP for
Research and
the IACUC Chair. If
you are handling
completion of this form, please contact the office at iacuc@uvm.edu
or call Ms. Abbey Peterson at 6-5040.
The office will assist in the accurate completion of the
form and in
obtaining institutional signatures.
A
sample form may be downloaded at http://grants.nih.gov/grants/olaw/sampledoc/
interinstitutional_assurance.htm.
7.D. Continuing Review and Six -Year Renewal
7.D.1. Requirements
There are requirements for continuing review in both the Public Health Service (PHS) Policy on Humane Care and Use of Laboratory Animals and the United States Department of Agriculture (USDA) Regulations on Animal Welfare. Animal welfare regulations require a continuing review at least annually for USDA-covered species and according to PHS Policy Section IV.C.5, “The IACUC shall conduct continuing review of each previously approved, ongoing activity covered by this Policy at appropriate intervals as determined by the IACUC, including a complete review in accordance with IV.C.1.-4. at least once every three years.” The IACUC developed the following processes to meet the requirements of both of the above. It is the policy of the IACUC to review all vertebrate animal research appropriate to the degree of pain involved, but not less than once per year. Each protocol continuing beyond every three-year point will receive complete de novo review (triennially).
If a
protocol remains open beyond a second triennial review, that
protocol will go through the 6-year renewal process which requires that
the PI
submit a new Animal Use Protocol form for that project. A new IACUC
protocol
number will be issued at that time and the previous protocol will be
effectively closed.
The purpose of continuing review is to monitor:
1) the status of the protocol
2) verify that completed activities were conducted in accordance with the approved protocols,
3) solicit information about activities projected for the coming year, and;
4) reflect changes in key personnel and whether mandatory training is complete
7.D.2. Annual and Triennial Continuing Reviews
For the first and second year of the protocol, the Investigator will be forwarded a notice and continuing review form for completion and submission. The Investigator will complete only the relevant sections and submit the form for review and continued approval.
Prior to the end of the third year the Investigator will be forwarded the same continuing review form indicating that it is a triennial review and that all sections need to be completed. The additional triennial section of the form allows the Committee to do a “complete review” as required by PHS Policy. The Investigator will also be provided a copy of his/her currently approved protocol. They will be required to confirm in the continuing review form that that is the protocol they are using. This will verify that the Committee and the Investigator’s records are in agreement.
For the fourth and fifth year of the protocol, the Investigator will be forwarded a notice of continuing review and a continuing review form for completion and submission. The Investigator will complete only the relevant sections and submit the form for review and continued approval. This is the same process used for the first and second year of the protocol.
Continuing
reviews that are not
on a triennial year will be reviewed administratively either
by the IACUC Chair (non-USDA regulated species only) or through the
Designated
Review Process.
Continuing
reviews that are on a triennial
year
will be reviewed by the designated review process to conduct a
“complete review.”
No
new changes to the
protocol
are to be requested by the Investigator at the time of continuing
review. Often, as
part of the completion of the
continuing review forms, it becomes apparent that a change in animal
numbers will
need to be made. If Investigators need to make a change in the
currently
approved protocol, they must submit a separate protocol amendment
request for
review. The same
reviewer will be
assigned the Continuing Review and any associated amendments whenever
possible.
As part of the continuing review process, the Committee may require that the research be restricted, modified, reviewed more frequently or terminated/suspended. Alternatively, special precautions or Committee-imposed restrictions, or shortened review periods, may be modified if current data support such actions.
The most visible element of the continuing review process approval is the Verification of Approval (VOA) form. This form indicates an expiration date which can only be extended through the Continuing Review process.
Expired Approvals: Extensions beyond the expiration date cannot be granted. If the expiration date has passed, or is in jeopardy of passing, the Committee must be notified as soon as possible.
a. If the Committee does not provide continued approval of the research by the specified expiration date, research activities are suspended and animals are moved to a holding protocol pending continued approval of the research by the Committee.
b. Researchers found to be conducting research activities without a current IACUC approval are in noncompliance with the regulations.
7.D.3. Six-Year Renewal
As
of 2018, protocols
will no longer be allowed to remain open for more than six years. If a
project
is ongoing, at the time that the second triennial review would be due,
the
protocol will be closed and the PI will be required to submit a new
protocol for
de novo review. The new protocol must reflect all current activities
being
conducted as well as any future plans. The new protocol will be
assigned a new
protocol number.
Protocols
that are due for six year de novo
review will be reviewed either
by full or designated review. The type of review depends upon the
anticipated
level of animal discomfort and types of procedures.
Prior
veterinary consultation and
scientific merit review is required for the six year de novo review. Protocols
requiring full committee review are
placed on the next available agenda, therefore it is important to
submit six-year
renewals minimally 6-8 weeks
prior to the expiration date to allow processing and review time. The
new
protocol must be approved before the old one expires or all animals
will be
moved to a holding protocol until the new protocol has received
approval.
To
assist researchers in meeting this requirement, one year prior to the
beginning
of the sixth year of the project, researchers will be forwarded a
specific
renewal notification. PIs will also receive a 120, 90, 60 and 30-day
reminder
prior to six-year expiration date.
PIs
must submit a current Animal Use Protocol form if they wish to extend
the project.
7.D.4. Documentation
Once approved, the Committee will return a signed Verification of Approval form to the PI via interoffice mail. A copy of the approved protocol is available to the Office of Animal Care and Use Management through the InfoEd electronic system. A copy of the VOA should be kept by the PI as documentation of continuing review completion.
7.E. Request for Modification/Amendment to Previously Approved Protocol
7.E.1.
Requirements
Review
of any changes to previously approved research is required by
regulation and is
an essential element of the ongoing review of research involving
vertebrate
animals. Regulations mandate that changes cannot occur until after
review and
approval.
The
IACUC recognizes that research is a continuous process and that changes
in the
conduct of the research are necessary. However, no changes to an
approved
protocol should be implemented until the IACUC has reviewed and
approved the
changes.
An
algorithm
has been developed
that categorizes amendments and shows the path of IACUC review that
each
category of amendment may take. The determination of whether a proposed
amendment is “significant” vs. “minor” falls to the IACUC chair, in
consultation with the University Veterinarian, as necessary.
7.E.2.
When
to Request
Requests
for approval of modifications may be submitted at any time. Complete an
Animal
Use Protocol
Amendment
form. The changes must be approved before any changes can be
implemented in the
conduct of the protocol. NOTE: If the amendment involves a new use of
hazardous
materials or radiation, additional review by other committees is
required prior
to implementation.
7.E.3.
How to Request
an Amendment to Approved Protocol and Review Process
The
Animal Use Protocol Amendment form is intended to capture all of the
required
elements for a significant review of proposed amendments. Complete the
form and
attach a revised version of the protocol.
Minor
amendments will usually be reviewed administratively by the IACUC Chair
and
Significant Amendments will be reviewed through the Designated Review
Process
in accordance with the algorithm noted above.
Note:
The Animal Use
Protocol Amendment form is located in the forms section of the IACUC
website
and should be downloaded each time it is needed as forms are frequently
updated.
Changes to protocol personnel require an amendment to the protocol. The Committee has a specific form, Request for Change in Personnel, to make these changes. Submit this completed form and your amended protocol. Note: Make sure any new personnel have completed the required training.
7.E.4.
Documentation
Once
approved, the IACUC will return a signed Animal Use Protocol Amendment
form.
Proof of the amendment approval must be kept by the PI (perhaps in a
Research
Regulatory Binder) as evidence that the IACUC has approved the change.
Note:
A copy of the approval is available to the Office of Animal Care and
Use within the InfoEd electronic system.
7.F. Notice of Termination
It
is the responsibility of the investigator to notify the IACUC when a
project is
completed. Projects that have been completed, withdrawn or terminated
are
closed immediately upon notification. The Office of Animal Care
Management is
notified by the IACUC of all closures. All animal use on a specified
protocol
is stopped. No further purchase of animals can be made under the
specified
protocol number.
8. PRINCIPAL INVESTIGATOR RESPONSIBILITIES
8.A. Expectations of a Principal Investigator
There are certain expectations of a principal investigator (PI). When the principal investigator submits a research protocol to the IACUC, by signing the Protocol Form you agree to the expectations listed below:
|
I
have provided an accurate description of the proposed animal use in
this protocol document. If the procedures concerning animal use in this
project are to be changed, I will obtain approval from the IACUC before
the changes are implemented. I
understand that failure to do this may place both me and the University
in violation of federal regulations and the Animal Welfare Act. In
addition to the above, I agree to the following conditions; ·
Correct
procedures for animal handling and restraint, administration of
anesthetics and analgesics, and AVMA recommended methods of euthanasia
are used in this project. ·
All
experiments involving live animals will be performed only by the
qualified individuals indicated in the personnel roster. ·
Personnel
involved in this project have been or will be adequately trained prior
to any animal work and will be given the opportunity to participate in
the University’s Occupational Health Program for individuals working
with animals in research or teaching. ·
Veterinary
care is provided promptly to any animals showing unanticipated signs of
pain or distress. ·
I
will make animal tissue available for sharing, or use tissues shared
from other protocols whenever possible. I
agree to abide by the U.S. Public Health Service Policy, the Animal
Welfare Act and University policies concerning the use of animals. As required by Federal
regulations, I confirm that the activities described herein do not
unnecessarily duplicate previous experiments, and that the animal
models proposed are the most appropriate for achieving the objectives
of this project. All
personnel will be informed that any concerns about the humane care and
treatment of animals or unlawful acts involving animals must be
reported to the IACUC Chair, the University Veterinarian, the Research
Protections Office, or to the Institutional Official using the
EthicsPoint™
website (https://secure.ethicspoint.com/domain/media/en/gui/24544/). Any individual reporting
such concerns cannot be discriminated against or subjected to any
reprisal. |
|
|
|
|
Other research personnel (technicians, graduate students and post-doctoral associates) have an equally important role in that they often conduct the day-to-day activities of the study.
8.B. Requirements of the Principal Investigator
As the principal investigator you must:
Ensure proper training and occupational health of the research team;
Ensure protocol adherence, and;
Provide reports on the progress of the study.
8.B.1. Ensure Proper Training of the Research Team
The principal investigator is responsible for ensuring that the research team has appropriate training prior to and during the conduct of the study as listed in Section 5.
8.B.2. Ensure Protocol Adherence
It is the principal investigator’s responsibility to ensure that the IACUC-approved protocol is being followed at all times by the research team. This includes making sure that amendments are submitted for IACUC review in a timely fashion and then once approved implemented by the research team.
8.B.3. Provide Reports on the Progress of the Study
During the course of a research study, new information might become available. As new information becomes available, the principal investigator is obligated to report to the IACUC. Common items that need to be reported in a timely fashion to the IACUC are described in detail below.
a. Continuing review of approved studies
It is the responsibility of the principal investigator to submit the current status of active protocols at least annually. Some protocols require more frequent review based on pain level/risk. The IACUC makes this determination and notifies the principal investigator. Please refer to Section 7.D. for additional information on submission of continuing reviews.
Note: As part of the progress report, investigators are requested to breakdown how the animals have been used by pain level. Therefore accurate records of ongoing animal use must be kept.
b. Reporting incidents and animal deaths
Principal investigators and/or research staff are responsible for reporting animal welfare incidents to OACM and IACUC. Please refer to Section 9 and the Reporting Animal Death Policy for additional information.
c. Amendments to a previously approved protocol
Requests for changes (amendments) to approved studies may be submitted at any time but before the change is implemented, it must receive IACUC approval. Refer to Section 7.E. for further instructions on how to submit an amendment.
e. Change in personnel
It is required that the investigator notifies the IACUC office when there are any additions or deletions to research staff participating in a protocol. Refer to Section 7.E.3. for further instructions on how to submit this type of amendment.
Amendments to add personnel will not be approved until the new personnel have completed the required training. Refer to Section 5 to learn more about required training.
f. Premature termination/suspension
The IACUC should be notified when a protocol has been terminated or suspended prematurely and the reasons for the premature termination or suspension. To notify the IACUC, submit a memo or email.
g. Study closures
Notify the IACUC when a protocol has been closed permanently.
8.B.4.
Coverage for PI (revised 5/5/15)
At
the time of initial protocol submission, the PI must either designate a
person
to be in charge and fulfill all responsibilities for oversight of the
protocol
in their absence or provide a plan for coverage (one option is to cease
activities during an absence – this requires IACUC notification). That person will
be identified in the
protocol as the designee. If
a formal
leave is planned, (e.g., sabbatical, medical, maternity or other
official leave
type) the IACUC needs to be notified, so that it may redirect protocol
inquiries
during that time. The
responsible
designee must understand the protocol and comply with the requirements
as noted
above.
8.C. Guidance for the Investigator
8.C.1. Communication with the IACUC
The
IACUC requires investigators to submit all
protocols and protocol-related submissions (e.g. amendments, key
personnel
changes) via email attachments.
Investigators in turn can expect to receive their IACUC
correspondence
via email. This
change is a giant step
forward and should result in less paperwork for the investigators and
the IACUC
staff.
We
continue to require protocol submissions to
be signed by the vet, the PI and others (e.g. departmental reviews) as
necessary. We have
identified a
potential pitfall with this new process to be confusion with document
versions. We must
all be vigilant about
making sure we are always working with the currently approved version
of the
protocol and protocol roster. Please
update your documents every time they are submitted by completing the
footer
with the date of the submission as shown below.

This
date footer is not automatic, therefore
you must change it each time you revise your
protocol. You
should not use the automatic date feature
as this will add further confusion by changing your date every time you
happen
to open the document. Failure
to update
this protocol version date may delay review of the submission.
All
submissions need to be sent to the
IACUC@uvm.edu
email box where new submissions will be
monitored and processed in the order they are received.
When you are in communication with the office, whether in writing, by telephone, fax or e-mail, you should have the following information available.
- IACUC number, if assigned at the time of contact
- Principal investigator’s name
- Protocol title
- Date and type of submission (if applicable)
We can more readily assist you with this information.
8.C.2. Written Communication of IACUC Decisions
Decisions made by the IACUC will be communicated to the principal investigator (or designee if provided) through a memorandum outlining the approval status and/or concerns, questions and/or comments of the IACUC. This correspondence will be forwarded via email to the principal investigator.
The IACUC notifies investigators and the institution of its decisions regarding protocol review through written memoranda and the minutes of IACUC meetings which are transmitted electronically. The decision to withhold approval is communicated to the investigator along with the reasons for withholding approval. There is no appeal process for the IACUC’s decision to withhold approval, however, an investigator may address the IACUC’s concerns by writing and submitting a new protocol.
The IACUC Chair will convey one of the following four decisions in writing to the investigator promptly after the meeting:
Approval
The principal investigator may begin the research study upon receipt of the Verification of Approval form.
Modifications
Required for Initial
or Continuing Approval
This decision is determined when the protocol is recommended for approval by the IACUC pending the investigator’s response to IACUC-directed stipulations/questions and/or revisions. The principal investigator must provide, via email, a memorandum responding to the IACUC’s recommendations. We ask that you indicate the IACUC number on this correspondence. If a revision to the protocol is necessary, attach a full revised protocol with changes indicated. Remember to update the protocol version date in the footer of the protocol.
Depending upon the issues that have been raised, the review of the response may occur through a Designated or Full process.
Withhold Approval
Questions regarding the scientific merit and use of animals are of such significance that the committee finds approval of the study to be unwarranted. The authority of the IACUC to withhold approval of a study may not be overridden.
NOTE: The IACUC has a 30, 60, 90 day reminder system for all pending protocol items. The investigator is reminded that the IACUC has requested something from them in regards to a protocol and is awaiting his/her response. At the 120 day mark the protocol is withdrawn from the Committee’s consideration. This helps to ensure that changes to protocols are handled in a timely fashion.
8.C.3. Accessibility of Records
The investigator must make available all research records for direct access by the IACUC staff. A copy of completed medical records for animals of USDA-covered species must be sent to OACM in a timely manner. Depending upon the protocol sponsorship there may be others with access needs such as the FDA or other regulatory authorities.
9. Animal Welfare Incident Reporting
9.A. Policies
9.A.1. In accordance with the Animal
Welfare Act, (9 CFR
Ch.1), Part 2 – Subpart C), 2.32. Training and instruction
of personnel include
methods whereby deficiencies in animal care and treatment are reported,
including deficiencies in animal care and treatment reported by any
employee of
the facility. No
faculty employee,
Committee members, or laboratory personnel shall be discriminated
against or be
subject to any reprisal for reporting violations or standards under the
Act.
9.A.2. Local Policy: Priority
One-UVM SOP #1.1.3 Reporting Animal Welfare Concerns
All procedures performed at the Priority One
Services, Inc. facilities and contract staffing services locations must
comply
with all applicable regulations governing the care and use of animals. Non-compliance will result
in corrective
action for the person(s) involved.
Any
concern regarding animal welfare will be taken seriously and
investigated. Corrective
action, if indicated will be taken
and the individual(s) expressing the concern will be informed.
9.B. Determining When and How
to Report Incidents
The
University has an online compliance reporting system “Ethics Point” (https://secure.ethicspoint.com/domain/media/en/gui/24544/index.html)
which can be accessed
through UVM’s Compliance & Privacy Services. This system allows
people to
anonymously report an animal welfare incident and receive information
about the
follow-up without compromising their identity. For those who prefer,
there are
also paper Animal Welfare Incident reporting forms available in the
central
Animal Facilities and available in an electronic format at the Office
of Animal
Care Management’s website (http://www.uvm.edu/~oacm/).
The General Animal
Care training outlines these methods of reporting animal welfare
concerns; in
addition, the AV gives many talks to students and staff throughout the
academic
year, describing the process of assuring that research animals are well
cared
for and the reporting process for animal welfare concerns.
10. OVERSIGHT AND MONITORING
10.A. Internal
Twice
each year the IACUC conducts a complete review of the University of
Vermont's
Animal Care and Use Program and inspects facilities where animals are
housed
and/or used. The NRC Guide for the Care and Use of
Laboratory Animals
and Animal Welfare Act and Animal Welfare Regulations
are the
principal documents used by the IACUC in its evaluations. Researchers who house
animals in their
laboratories over 12 hours should expect visits by the subcommittee of
the
IACUC at approximately 6-month intervals. All
survival and non-survival surgical sites
are also visited. Researchers
can expect
a notice of inspection 1 month in advance.
Written
reports of the program
evaluation and inspection are prepared according to PHS policy criteria
and are
submitted to the full committee for discussion and modification, if
necessary,
prior to taking action. Final
reports
are then forwarded to the institutional official.
In
addition to looking at the research facilities during the semiannual
inspection, IACUC members will conduct protocol monitoring visits. For further information, see
Monitoring
Process/Protocol Follow-up policy.
10.B. External
Annually
an Animal Welfare Officer from the USDA will inspect the animal
facilities and
may inspect individual labs. The
inspector will meet with the University Veterinarian or a designee who
will
escort the Animal Welfare Officer through the facilities.
Attachment A Committee Policies
These are direct links to the
individual policies that
have been approved by the IACUC.
|
# |
Policy
|
Date Reviewed |
Date Originally Approved |
|
1. |
10/22/18 |
01/12/01 |
|
|
2. |
10/22/18 |
10/22/18 |
|
|
3. |
NA |
12/17/07 |
|
|
4. |
Antibody
Production (retired 11/20/12) |
inactive |
04/28/03 |
|
5. |
Autoclave
Use and Sterilization (05/03/11 refer questions
to EHS) |
inactive |
11/24/03 |
|
6. |
09/18/18 |
3/24/08 |
|
|
7. |
Biological
Agents (included in Hazardous Material Policy) |
inactive |
inactive |
|
8. |
03/19/18 |
02/24/03 |
|
|
9. |
01/28/19 |
10/23/06 |
|
|
10. |
09/25/17 |
12/22/03 |
|
|
11. |
09/25/17 |
02/28/05 |
|
|
12. |
09/25/17 |
07/11/05 |
|
|
12.1 |
9/26/16 |
03/28/11 |
|
|
13. |
03/19/18 |
05/28/03 |
|
|
14. |
Frog
Oocyte Harvest (retired 01/28/19) |
inactive |
11/24/03 |
|
15. |
Housing
Animals Outside and/or Removal from Central Animal Facility |
11/19/18 |
12/22/03 |
|
16. |
03/19/18 |
04/28/03 |
|
|
16.1 |
01/28/19 |
11/23/09 |
|
|
17. |
Occupational Health and Safety
Program (section 5.D) |
02/25/19 |
10/23/06 |
|
18. |
Ordering
Animals |
inactive |
inactive |
|
19. |
Pain
and Distress, Determination of Levels |
05/21/18 |
05/28/03 |
|
20. |
Pet
Policy (refer to University policy on pets) |
inactive |
inactive |
|
21. |
05/21/18 |
05/28/03 |
|
|
22. |
12/05/18 |
01/23/06 |
|
|
23. |
09/17/18 |
11/28/05 |
|
|
24. |
07/18/17 |
05/28/03 |
|
|
25. |
04/22/19 |
02/24/03 |
|
|
25.1 |
01/23/17 |
1/24/11 |
|
|
26. |
12/05/18 |
05/18/09 |
|
|
27. |
12/05/18 |
01/23/06 |
|
|
27.1 |
05/22/17 |
10/22/07 |
|
|
28. |
Votey
Satellite Facility, Oversight Plan for |
inactive |
inactive |
|
29. |
01/23/17 |
11/28/05 |
|
|
30. |
05/21/18 |
01/23/12 |
|
|
31. |
05/21/18 |
2/27/12 |
|
|
32. |
11/28/16 |
2/25/13 |
|
|
33. |
03/26/18 |
03/26/18 |
|
34. |
04/22/19 |
04/22/19 |
Committee Forms
All
forms and form
instructions are located in the forms section of our website and should
be
downloaded each time you need one. (SEE: http://www.uvm.edu/iacuc and click on “All Forms”)
Attachment C: Animal Use Amendment Flow Chart
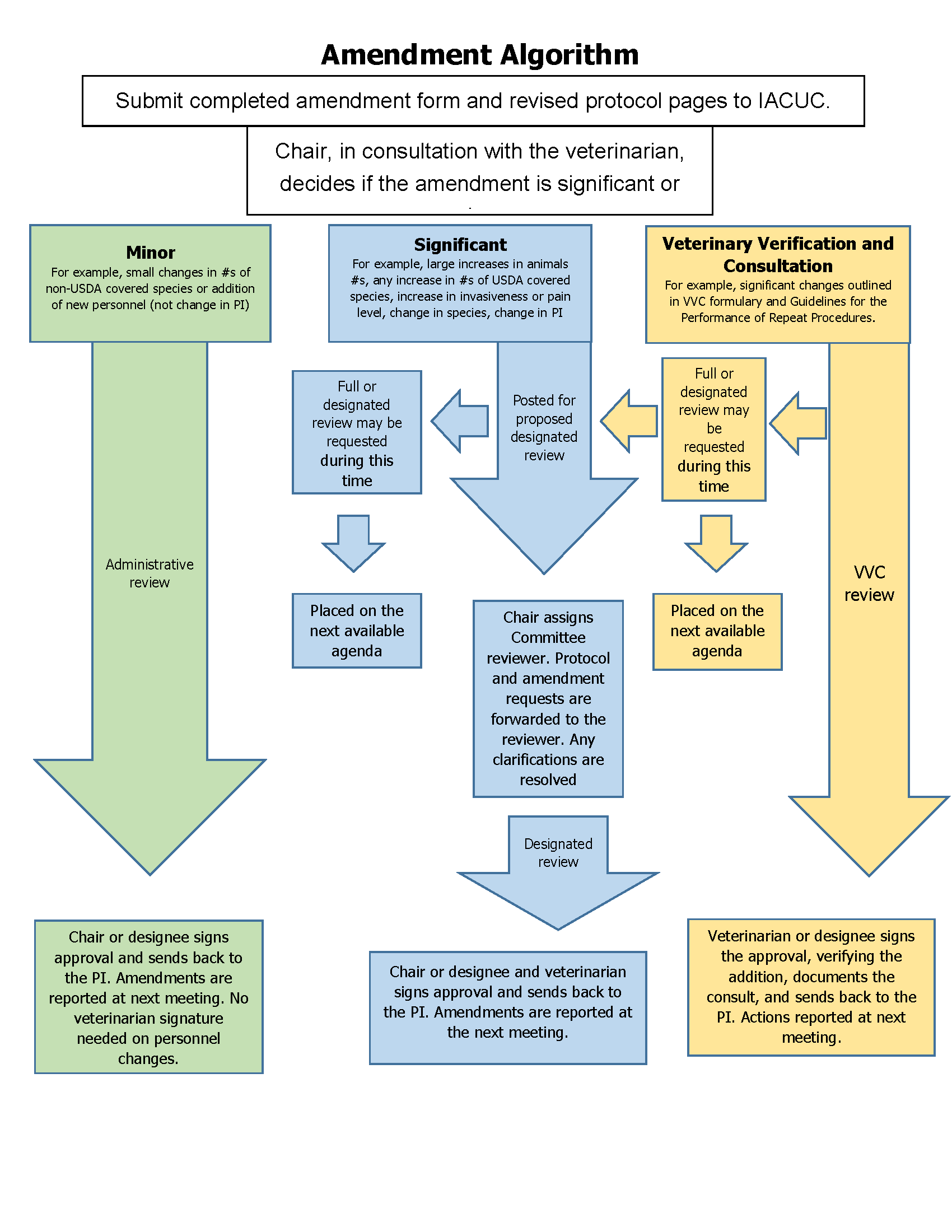
Attachment
D: IACUC NONCOMPLIANCE POLICY AND PROCEDURES
OVERVIEW
It
is the responsibility of the Institutional Animal Care and Use
Committee (also
referred to as IACUC) in accordance PHS Policy.IV.B.1-8 and 9 CFR.2.31
(c)
(1)-(8) and 2.31 (5) (6) & (7) to review reports of
noncompliance with
animal care and use within the institution.
To exercise this authority the IACUC is empowered to
inspect
laboratories, procedure areas, animal housing areas, and to sequester
research
or training records. The IACUC may receive reports in several different
ways
including external complaints, internal complaints, Animal Incident
Reports,
random and directed site visits, and investigator self-reporting. The
IACUC
encourages faculty, staff and/or students to report instances of
noncompliance,
especially when animal or human health and welfare is in question.
This
document describes the procedures for handling these matters. This policy is not all
encompassing, and the
IACUC reserves the right to use its discretion in individual cases.
DEFINITIONS
Noncompliance
is defined as the
conduct of research in a manner that deviates from the approved
protocol or
disregards or violates federal regulations or institutional policies.
Noncompliance may result from actions or omissions by study personnel,
and can
range from relatively minor or technical deviations to serious
deviations that
threaten animal welfare.
Serious
Noncompliance
is defined as noncompliance that, in the judgement of the IACUC,
potentially
increases the risk of harm to animals.
Continuing
Noncompliance
is defined as a pattern of noncompliance (recurring or ongoing) that,
in the
judgement of the IACUC, may indicate an underlying deficiency in
knowledge of
the regulations or IACUC requirements or an unwillingness or inability
to
comply with these regulations/requirements.
General
Noncompliance Review Procedures
The
investigation of potential noncompliance begins when the IACUC becomes
aware of
potential noncompliance. This may include an allegation (unproved
assertion) of
noncompliance, a self-disclosure of noncompliance, or any other
indication that
noncompliance may have occurred. The
process for the review of potential noncompliance involves initial
administrative review, followed by an inquiry/fact finding process if
indicated. Once
complete, the IACUC
makes a determination as to whether the noncompliance is serious,
continuing,
or neither. The IACUC determination will be documented in a summary
report that
contains a corrective action plan in cases of serious or continuing
noncompliance. This process is detailed below, however at any point in
the
review process, the IACUC designee may at their discretion:
·
Recommend
intervention for the safety of animals
·
Recommend
the suspension of research activities
·
Inform,
involve, and/or provide salient documents to the PI, members of the
research
team, the Department Chair, Dean, legal counsel, or Institutional
Officials, as
appropriate
·
Initiate
reporting per federal regulations
·
Initiate
a monitoring visit
·
Recommend
immediate corrective actions
Process
of Noncompliance Review and Determination
Initial
Review of Allegation or Indication of
Noncompliance: When
there is an allegation or indication of noncompliance, the first step
is an
administrative review to determine if, in the judgement of the
person(s)
conducting the review, there is the potential for
serious or continuing
noncompliance. The initial review may be conducted by the RPO Director,
RPO
Assistant Director(s), an IACUC Chair (Associate Chair or Chair),
University
Veterinarian or another Institutional Representative.
Allegations/indications
which are determined to have no potential to be
serious and/or
continuing noncompliance are resolved with either no follow-up (i.e.
when an
allegation or indication has no merit) or directly with the PI.
Inquiry/Fact
Finding Process:
If it is determined that the noncompliance has the potential
to be
serious or continuing or if questions remain following the initial
review, then
an inquiry (fact finding) process will begin.
The particular circumstances of the noncompliance will
determine when
the fact finding begins and when the committee is briefed. The fact
finding may
be conducted by any IACUC designee including a sub-committee or
subcommittee
member, the RPO Director, Assistant Director(s), an IACUC Chair
(Associate
Chair or Chair) or other Institutional Representatives. The IACUC may
be
briefed at any point throughout the fact finding process, as deemed
appropriate
by the designee. The fact finding process continues until the designee
has
arrived at a recommendation of determination (i.e. serious
noncompliance and/or
continuing noncompliance, or neither). A fact finding report is then
prepared
and includes the recommendation of determination and draft corrective
actions. This fact
finding report will
be shared with the PI, and if applicable, other person(s) involved. All parties will be
provided an opportunity
to respond to any factual inaccuracies within the report before the
committee
deliberates.
Deliberation
by
the IACUC:
At a convened meeting, the IACUC will consider all available
information and
make a determination as to whether the fact finding revealed serious
noncompliance and/or continuing noncompliance, or neither. The
following
factors will be taken into consideration by the IACUC or designee in
making
their initial determination as to whether the noncompliance is serious
and/or
continuing noncompliance. As each situation is unique, the indicators
of
noncompliance that are important in one case may not be relevant in
other
cases.
Factors
in the Determination of Serious
Noncompliance:
·
Level
of risk or potential risk to animals
·
Severity
of violation of the research process
·
Frequency
or number of minor deviations or errors
·
Intent
·
Threat
to integrity of the IACUC review processes and requirements for the
protection
of animals (i.e.
falsification of IACUC
documents)
·
Other
factors that, in the judgement of the IACUC or designee, are relevant
to the
situation being reviewed.
Factors
in the Determination of Continuing
Noncompliance:
·
Similarity
of noncompliance to previous deviations and/or noncompliance within the
same
protocol.
·
Similarity
of noncompliance to previous deviations and/or noncompliance in other
protocols
conducted by the investigator.
·
Likelihood
that instances of noncompliance will continue without intervention
Final
Determination of the IACUC: If,
in the judgement of the committee, the
noncompliance is neither serious nor continuing,
this determination will
be shared with the PI. If, in the judgement of the committee, the
noncompliance
is serious and/or continuing, the designee will
prepare a summary report
including the IACUC’s determination and an approved corrective action
plan. This report
will be shared with
the PI, who will be given 14 days to review it before it becomes final.
Development
of Corrective Action Plans:
The
IACUC/designee will develop a proposed
plan for corrective actions based on the information gathered during
fact-finding and input from the principal investigator and/or other
affected
individuals. The proposed plan may:
·
Require
no further action
·
Require
minor corrective actions to achieve compliance
·
Require
additional education
·
Require
the investigator and/or other affected individuals to develop and
implement
procedures to prevent recurrence
·
Review
internal departmental or institutional mechanisms and systems for
opportunities
to prevent recurrence or similar occurrences by others
·
Require
additional oversight (e.g., by other faculty member or department
process)
·
Require
more frequent IACUC reviews
·
Require
internal monitoring visits or monitoring plans
·
Suspend
or terminate individual protocols
·
Restrict
researcher’s research activities
REQUESTS
FOR RECONSIDERATION
A
PI may request a reconsideration of the IACUC’s determination. Requests
must be
limited to claims that either (1) the process was faulty, resulting in
considerable risk that the outcome was incorrect; or (2) that the
findings
and/or corrective actions imposed by the IACUC were excessive or
unjustified. The
written request must be
submitted within 14 days of receipt of the summary report and must
specify the
nature of any claimed procedural error or the perceived unfairness of
actions
taken. Reconsiderations will be conducted by an IACUC Chair (Chair or
Associate
Chair), or Designee. The
reconsideration
process will result in one of three outcomes, either the summary report
will
stand and it will become final, the summary report will be modified and
become
final, or further investigation is necessary and will be initiated.
Required
reporting
When
noncompliance is determined to be serious and/or continuing, the final
report
will be forwarded to federal regulators if required, and to applicable
Institutional Officials, the Departmental Chair, the Dean, and
sponsors, if
applicable.
Guiding
Principles for NonCompliance Review
Protection
of Animals: The
University of
Vermont is committed to the humane care and use of animals in
activities
related to research, testing and teaching.
Fairness: The IACUC
strives to maintain a review that
is impartial and honest, free from self-interest, prejudice or
favoritism.
Communication:
The committee
will communicate with the PI during the review process at points
determined to
be appropriate by the IACUC designee.
Confidentiality:
All IACUC
discussions and documents regarding a situation of noncompliance are
considered
sensitive and will be handled in a confidential manner and in
accordance with
state and federal regulations. The
IACUC
cannot, however, guarantee complete anonymity to informants or
witnesses. Confidentiality
will be maintained to the
extent possible to protect privacy and prevent retaliation, while still
allowing for a full and fair review. Information may be shared, as
described
above under Required Reporting.
Conflict
of Interest: Any IACUC
member who feels that they have a conflicting interest must recuse
themselves
from reviewing the issue of noncompliance.
IACUC members who are also listed as key personnel on the
protocol(s)
will not participate in the review but may be asked for information.
Procedures: In addition to what has
been stated within
this policy, the Committee will follow all applicable procedures that
are
outlined within the Committee Operating Procedures document.
Attachment
E: Performance of Repeat
Procedures
(1 Adapted from Colorado State University https://vpr.colostate.edu/RICRO/IACUC November 18, 2016)
Background
Proper
use of animals, including the avoidance or
minimization of discomfort, distress, and pain while simultaneously
minimizing
the numbers of animals needed when consistent with sound scientific
practices,
is imperative1. Furthermore, the Guide2 specifies
that the Institutional
Animal Care and Use Committee (IACUC) should weigh study objectives
against
animal welfare concerns in accordance with the tenets of the Three R’s.
The
IACUC is often confronted with a situation where procedures may have to
be repeated
in an individual animal in order to achieve study objectives. This
document
discusses examples of common scenarios that can be used as a roadmap to
IACUC
decision making. Regardless of the procedure to be performed there
should be
appropriate justification as to why a procedure needs to be repeated,
an
indication of the interval between the repeats as well the total number
of
repeats together with a statement of the anticipated long term effects
of the
repeated procedures on the animal clearly articulated in the protocol.
The
total numbers of procedures that can be performed is dependent on the
skills of
the individual performing it, the nature of the procedure, other
procedures
previously performed on the animal, future procedures that may need to
be performed,
the temperament of the animal, and health as well as physiological
status of
the animal.
The
changes within this document must be handled in
compliance with the VVC process described in NOT‐OD‐14‐
126 and in the IACUC reviewed
and approved Protocol Review Process document.
Procedures
and guidance
Oral
gavage
Oral
gavage is a widely used method for safely administering
known quantities substances to animals by properly trained
(experienced/qualified)
personnel. Oral gavage is part of accepted routine toxicological
testing
accepted by agencies like Food and Drug Administration and Organization
for
Economic Cooperation and Development’s 28 or 90-day oral toxicity,
where
substances are administered daily during that time period. The maximum
dosing
volume is 1.5 ml for a 30-gram mouse (10 ml/kg), and 16 ml for a
400-gram rat
(10‐20
ml/kg)3. When performed properly, the procedure can be used to
administer 50‐200
μl into the stomach
of mice daily for up to 20 weeks.
Fluid
collection
Studies
often require repeated (timed) collections of fluid
e.g. blood, cerebrospinal fluid, rumenal fluid, urine, synovial fluid,
intraocular fluid, semen, milk, tracheal wash, broncho‐alveolar
lavage, etc. In some
rare instances this can be achieved by “free catch” such as for urine
or
collecting feces after defecation, however, for the most part the
process
requires penetration of a cavity typically with a needle, cannula,
catheter or
trocar with or without anesthesia and/or analgesia or even use of an
assistive
device e.g. electro‐ejaculator
or artificial vagina. Most of these procedures are relatively benign
and pose
little or any adverse impact when done correctly by a properly trained
(qualified or experienced) individual. Animals should be monitored
during and
after the
procedure, and be provided appropriate analgesia or anesthesia. The
veterinarian should be notified in case of development of complications
to
facilitate resolution of the problem.
The
circulating blood volume for most common laboratory species is 55‐70
ml/Kg (5‐7%
of the body weight). The value is influenced
by the status of the animal including age, body condition and health.
Up to 10%
of the total blood volume (1% of the body weight) can be safely removed
in a
normal healthy animal. This volume may be collected once in 3‐4
weeks4. This period allows the animal to recover from the potential
adverse
effects of blood loss. Again the same 3‐4
week recovery period
should be followed. If you are collecting 0.75% of the body weight a
recovery
period of 2 weeks may be sufficient, and for 0.5% a week’s recovery is
okay and
for 0.05% blood can be collected daily. The same total amount of blood
can also
be removed as multiple quantities over a 24 hour period. If necessary
fluids
can also be administered to support the blood volume, especially for
collections at the upper end of the limit.
The
table below provides examples of maximal
amounts of blood that can be collected.

The
table below provides common sites for blood collection in a number of
laboratory animal species.

The
following table indicates the common routes for administration of
substances.6
Suggested maximum volumes to be administered are shown in parenthesis.
For most
mammalian species these volumes can be safely administered: 5‐20
ml/kg orally; 5 ml/kg subcutaneously; 0.05 ml/kg intramuscularly; 10
ml/kg
intraperitoneally; and 5 ml/kg intravenously as a bolus or 2‐4
ml/kg continuous infusion.7

Substances
to be administered should be sterile and if possible the pH should be
near
neutral.
*Note
that intramuscular injections in mice are discouraged unless
scientifically-justified.
Rumen
fluid is sometimes collected for research or clinical purposes. The
rumen of an
adult dairy cow contains 184 liters (49 gallons) of fluid. An adult
sheep or
goat rumen is about 3‐6
gallons (11.4 – 22.8 liters). Rumen fluid can
be collected either through a permanent fistula or using a stomach.
Creating of
a fistula makes it easy to collect rumen fluid and is usually the
preferred method
for long term repeated fluid collection. The process requires surgery
to create
the fistula as well as regular cleaning and maintenance of the fistula.
Use of
a stomach tube is fairly straight forward, however, it causes a
temporary
discomfort to the animal, and has the potential to cause esophageal and
oropharyngeal irritation or trauma, and the potential for aspiration
pneumonia.
Use of a stomach tube does not require surgery and a recovery period.
Rumen
fluid can be collected repeatedly by a trained or experienced
individual
provided due care is take to avoid the potential adverse impact of the
procedure e.g. alteration of rumen microbiome, dehydration, digestive
disturbances, trauma and/or aspiration pneumonia especially if a
stomach tube
is used. When 5 or more gallons of rumen fluid is removed daily, it is
necessary that the fluid be replaced with water.
Aseptic
technique and appropriate pain management are required for collection
of
synovial fluid (arthrocentesis). The amount of fluid that can be
collected will
be dependent on the joint cavity as well as the species. The knee joint
often
provides the most accessible and largest practical volume of fluid. The
amount
of fluid in the joint is typically very small. The human knee joint
contains
about 4 ml of synovial fluid. The equine stifle joint has about 20 ml
of
synovial fluid and the carpal joint has about 10 ml. For
pharmacokintetic
studies arthrocentesis can be done at 0, 6, 12, 24 and 72 hours and can
be
repeated with a rest of 1‐2
weeks (Frisbie, 2014)11. Arthrocentesis can
be safely performed every week for 10‐20
weeks.
Cystocentesis
should be performed after cleaning the puncture site with antiseptic
solution.
If urine is not obtained at the first attempt change the needle before
making
the second attempt. The procedure should be aborted after the third
unsuccessful attempt. The needle should not be redirected during the
procedure.
Cystocentesis may induce mild transient hematuria, bruising and urine
leakage. Healthy
bladders heal relatively quickly. It is possible to collect urine daily
by
cystocentesis especially if the procedure is ultrasound guided which
minimizes
the possibility of trauma (Lappin, 2014)12.
Nasal
swab and buccal swabs can be performed non‐invasively
using a
sterile cotton tip applicator, or similar device. They can typically be
conducted with manual restraint. If the animal is uncooperative, then
the
procedure should be aborted. Nasal and buccal swabs can be collected up
to 3
times a day. As long as there is no evidence of trauma such as nose
bleed,
these swabs can be collected up to 30 consecutive days.
Saliva
can be collected from the oral cavity. If the animals are conditioned
to
produce saliva using training methods as described in the protocol,
saliva can
be collected up to 3 times daily. If medication is needed to collect
saliva, it
should be given no more than once daily.
Feces
can be collected up to 3 times daily by manual collection from animals
larger
than rabbits using a lubricated fecal loop. Voided feces can be
collected as
often as desired.
![]()
1 United States Government
Principles for
the
Utilization and Care of Vertebrate Animals Used in
Testing, Research,
and
Training.
Principle
III and IV.
http://grants.nih.gov/grants/olaw/references/phspol.htm#USGovPrinciples
2 Guide for the Care
and Use of Laboratory Animals (2011), National Research
Council, National
Academy Press,
Washington, DC. P27‐28.
3 Diehl KH, Morton R, Morton D, et al
(2001).
A good practice guide to
the administration of
substances and
removal of blood including
routes
and volumes.
Journal
of Applied
Toxicology
50(5): 600‐613.
4 Diehl KH, Hull R, Morton
D, et al
(2001). A good practice
guide to
the
administration of
substances and removal
of
blood,
including routes
and volumes.
Journal of
Applied Toxicology 21: 15‐23.
5 Adapted from Joslin
JO (2009)
Blood collection techniques in exotic small mammals.
Journal of Exotic Pet
Medicine 18(2): 117‐139.
6 Adapted from Diehl KH,
Hull R, Morton D, et
al (2001). A good practice guide to
the
administration of
substances
and
removal of
blood, including
routes
and volumes. Journal of Applied Toxicology 21: 15‐23.
7 Turner PV, Brabb T, Pekow C, Vasbinder MA (2001).
Administration of
substances
in laboratory animals: routes of
administration
and factors
to consider. Journal of the American Association
for Laboratory Animal
Science 50
(5): 600‐613.
9 Koch MA (2006)
Experimental modeling and research
methodology. Chapter 18, P
606‐607. Suckow MA,
Weisbroth SH,
Franklin CL
(eds.).
2nd edition. The
Laboratory Rat. Academic
Press/Elsevier,
Amsterdam.
10 Iwarsson K, Lindberg
L, Waller T (194) Common
non‐surgical techniques and procedures.
Chapter
16, P267.
Svendsen P, Hau J
(eds.)
Handbook of
Laboratory Animal Science. Volume
1. CRC
Press,
Inc, Boca
Raton, FL.
11 Frisbie D (2014). Personal communications. 12 Lappin, M
(2014). Personal communications.
