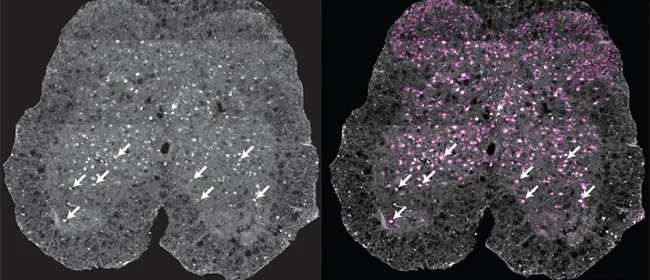
A biological mechanism that leads to neuron cell death in people with multiple sclerosis (MS) can be blocked with gene therapy and new drugs currently under development in the laboratory. This finding points to potential treatments for progressive MS, which is currently untreatable.
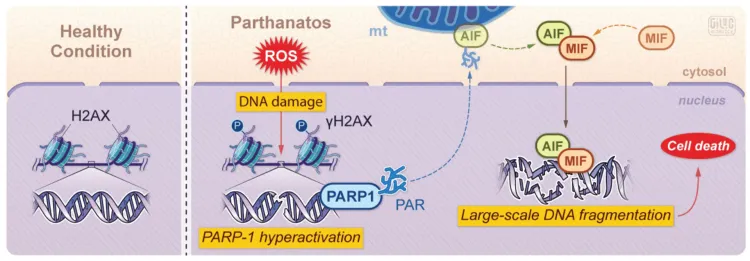
This research study at the University of Vermont (UVM) Larner College of Medicine focused on a specific molecular process that leads to cell death, called parthanatos, which occurs in autoimmune diseases that cause inflammation of neuronal brain tissue, including MS. The findings, published in Nature Neuroscience, suggest that damage to neurons associated with MS is caused by immune responses that trigger parthanatos, and that this trigger can be prevented.
“The fact that you can intervene downstream of the inflammation and rescue the cells as they are starting to die is really exciting. This has never been tried before,” said senior author Peter A. Calabresi, M.D., professor and chair of the UVM Department of Neurological Sciences. “This could be a good strategy for treating progressive MS and related neurodegenerative diseases.”

MS is the most common cause of neurologic disability in people under age 60. Nearly one million people in the U.S. live with M.S., and in northern regions including New England, MS affects up to one in 500 people. Prevalence of MS is rising, according to the National Institutes of Health, and women are more likely to have MS than men.
The disease causes the body’s immune system to mistakenly attack myelin, the protective sheath surrounding nerve fibers. These attacks cause inflammation and lesions on the spinal cord and brain, disrupting electrical signals connecting the brain and the body. Symptoms range from muscle weakness, numbness, and fatigue to problems with balance and vision, as well as mental difficulties. For most people with MS, the attacks come and go at unpredictable intervals, which is referred to as relapsing-remitting MS.
“Previous research focused on how to block the immune system from attacking the myelin, and we now have more than 25 FDA-approved treatments for that. But these treatments do nothing to repair the lesions that form from early damage to the myelin. The lesions cause a slow, insidious neurodegeneration in the brain and spinal cord,” Dr. Calabresi said. The progressive damage can lead to life in a wheelchair, loss of vision, and full debilitation.
He likened this process to what happens to a tree that loses a branch. The tree is fine for a while, but the stump where the branch came loose slowly rots, gradually killing the tree.
“The typical person who presents with MS in their 20s will go on treatment and their MS quiets down until they are about 40, when they may start to have a slow worsening of the symptoms they initially presented with,” he said. For example, “if they had a numbness and weakness in their leg at onset, by age 40 they often aren’t able to run or walk distances, and by age 45 they may walk with a limp, and by age 50 they are using a cane” due to the slow neuron cell death at the lesion site.
“The fact that you can intervene downstream of the inflammation and rescue the cells as they are starting to die is really exciting. This has never been tried before.” — Peter A. Calabresi, M.D.
Using models and brain and spinal cord tissue from people with MS, Calabresi’s laboratory team assessed DNA inside neurons, looking for signs of damage. They found molecular markers of parthanatos. While parthanatos had been previously identified in the progression of neurodegeneration in Parkinson’s and Alzheimer’s disease, Calabresi’s team is the first to report that parthanatos is responsible for a sizable portion of cell death in MS.
The team then introduced a mutant gene that blocked the immune response that triggers parthanatos. They also successfully inhibited the parthanatos process with a small molecule drug. This work could eventually inform the development of new pharmaceuticals or genetic interventions that prevent the death of brain cells in MS, in turn reducing progression of the disease and severity of symptoms.
The next steps in this research, which is funded by the U.S. National Institutes of Neurological Disorders and Stroke and the U.S. Department of Defense, are to replicate the findings at different points in MS disease progression and work with medicinal chemists to develop compounds that are safe and effective for long-term use in people.
“Translational research is a long process,” said Calabresi, who has been working on this study for five years and recently filed for a renewal of the R01 application that funded it. He has studied therapeutic targets on immune cells for more than 30 years, starting with a fellowship at the National Institutes of Health and appointments at the University of Maryland School of Medicine and Johns Hopkins School of Medicine. Calabresi joined UVM Larner College of Medicine in October 2025 and will soon be joined by his laboratory staff from Johns Hopkins.
Read the study in Nature Neuroscience
Research like this has contributed to the University of Vermont’s designation by the Carnegie Classification of Institutions of Higher Education as an R1 institution, placing it in the top tier of research universities in the U.S.