At UVM, you’ll have access to top scholars and courses, and you’ll live and study alongside friends with diverse passions and majors whose ideas and interests will expand your world. With this deep and broad education, you’ll launch prepared not only to land a great job, but with the ability to see the world through multiple lenses, an essential 21st century life skill.
CNHS offers undergraduate, graduate, and certificate education in Communication Sciences and Disorders, Exercise Science, Integrative Health, Medical Laboratory Science, Medical Radiation Sciences, Nursing, Occupational Therapy, Physical Activity and Wellness, Physical Therapy, Speech-Language Pathology, and Public Health.

Discover. Teach. Heal.
At the UVM College of Nursing and Health Sciences (CNHS), students learn to reach across divides for the benefit of individuals and communities, to become leaders among the next generation of culturally sensitive health professionals.
In and beyond the classroom
Academics
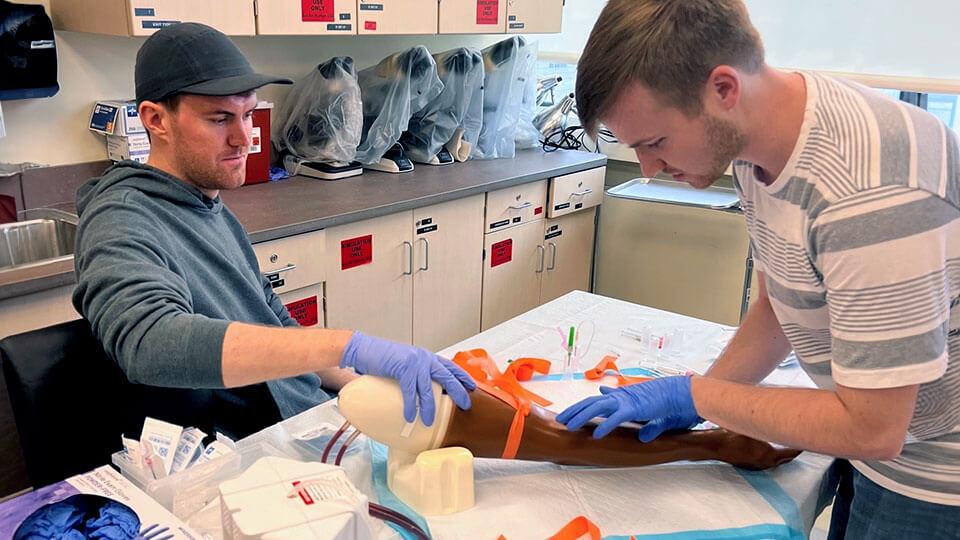
Classroom
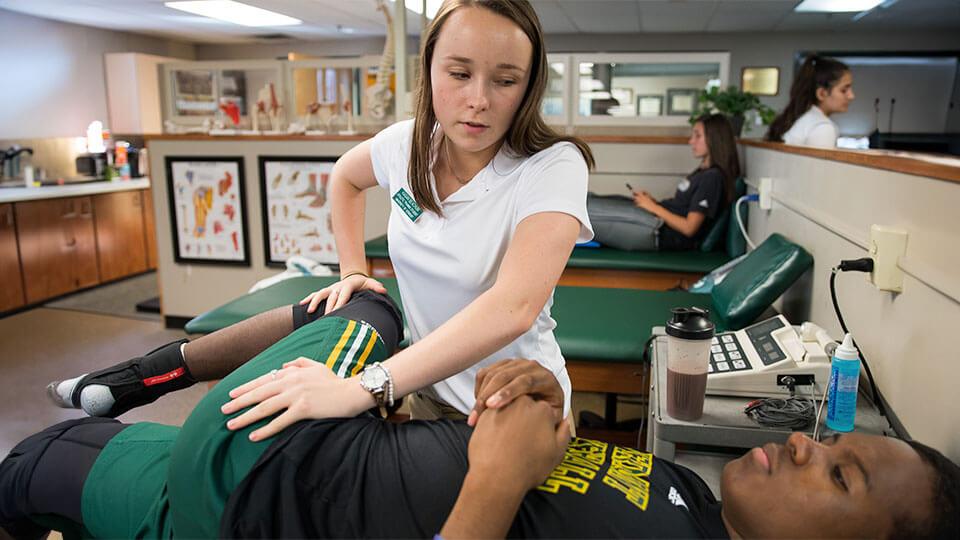
Practice

Research
By the numbers
- 1048 Undergraduates
- 9 Graduate Degree Programs
- Top 10% of Nursing Programs (U.S. News 2024 Rankings)
- 201 Graduate Students
- 4 Academic Departments